G. Marius Clore, M.D., Ph.D., FMedSci, FRS, NIH Distinguished Investigator

Professional Experience
- NIH Distinguished Investigator, NIDDK, NIH (2011-present)
- NIH Senior Investigator, NIDDK, NIH, 1988-present
- Head, Biological NMR Group, Max Planck Institute for Biochemistry, Martinsried, Germany, 1984-1988
- Member of Scientific Staff, MRC National Institute for Medical Research, London, U.K., 1980-1984
- House Surgeon, St. Charles Hospital (St. Mary's Group), 1980
- House Physician, University College Hospital, 1979-1980
- Ph.D., MRC National Institute for Medical Research, London, 1982
- M.D., University College hospital Medical School London, 1979
- B.Sc. (1st class honors), University College, London, 1976
Research Goal
The purpose of my lab’s research is to understand the interrelationship between the structure, dynamics, and function of proteins. A particular focus is the study of rare, highly transient, “excited” states of proteins and their complexes that play a key role of molecular recognition.
Current Research
Our lab studies the structure and dynamics of proteins, protein-protein complexes, and protein-nucleic acid complexes using multidimensional nuclear magnetic resonance (NMR) spectroscopy, and we develop and apply novel NMR and computational methods to aid in these studies. We are particularly interested in complexes involved in signal transduction and transcriptional regulation, and on AIDS and AIDS-related proteins. More recently we have focused on the development of novel NMR methods to detect, visualize, and characterize transient, sparsely-populated states of macromolecules. Such states, which are invisible to conventional biophysical techniques, including crystallography, play a critical role in macromolecular recognition, allostery induced fit, conformational selection, and molecular assembly. Dr. Clore is a Fellow of the Royal Society, a Member of the National Academy of Sciences, a Fellow of the U.K. Academy of Medical Sciences, a Fellow of the American Academy of Arts and Sciences, and a Foreign Member of the Academia Europaea. Prizes and awards include the Royal Society of Chemistry Centenary and Khorana Prizes, the Biochemical Society (U.K.) Centenary Award, The Biophysical Society Innovation Award, and the Biopolymers Murray Goodman Memorial Prize.
Applying our Research
This research will facilitate targeted and rational drug design.
Select Publications
- Nucleation of Huntingtin Aggregation Proceeds via Conformational Conversion of Pre-Formed, Sparsely-Populated Tetramers.
- Torricella F, Tugarinov V, Clore GM.
- Adv Sci (Weinh) (2024 Jun) 11:e2309217. Abstract/Full Text
- Time-resolved DEER EPR and solid-state NMR afford kinetic and structural elucidation of substrate binding to Ca(2+)-ligated calmodulin.
- Schmidt T, Jeon J, Yau WM, Schwieters CD, Tycko R, Clore GM.
- Proc Natl Acad Sci U S A (2022 Feb 8) 119. Abstract/Full Text
Research in Plain Language
Our lab is developing new tools and techniques that facilitate the study of the structure and dynamics of proteins and protein complexes, functional units that include one or more proteins. Our studies rely on nuclear magnetic resonance (NMR) spectroscopy, a research approach that relies on the magnetic properties of the nucleus of certain atoms to determine physical and chemical properties of the molecules in which they are contained.
Currently, we are especially interested in developing novel NMR approaches to detect and visualize short-lived, sparsely-populated states that are invisible to conventional biophysical and structural techniques. Such species play a critical role in recognition and molecular assembly. Other interests include the development of hybrid strategies to solve the structures of large (> 100 kDa) complexes using a combination of NMR and solution X-ray scattering techniques.
Our research extends the use of NMR to studies that were previously impossible. For instance, NMR can be used for larger structures. The many proteins and protein complexes we describe are resulting in new insights into fundamental cell operations. Our many advances in the use of NMR also include the development of mathematical algorithms and computational techniques that are making analysis of NMR data faster and more efficient.
Related Links
- Dr. Clore's Curriculum Vitae (PDF, 1 MB)
- National Academy of Science Profile
- Academia Europaea (The Academy of Europe) Profile
- Wikipedia listing
- Royal Society Profile
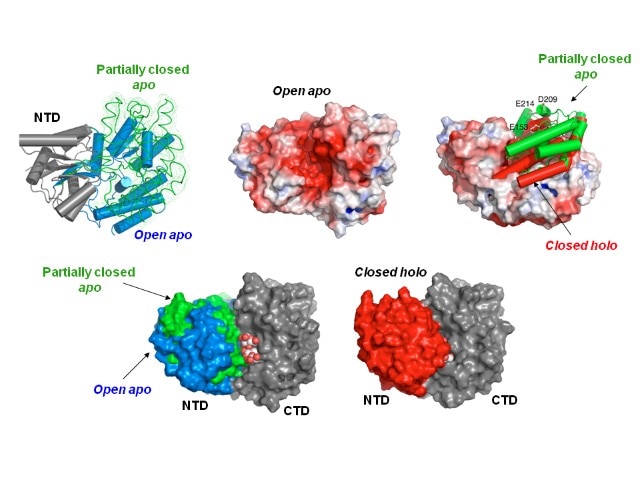
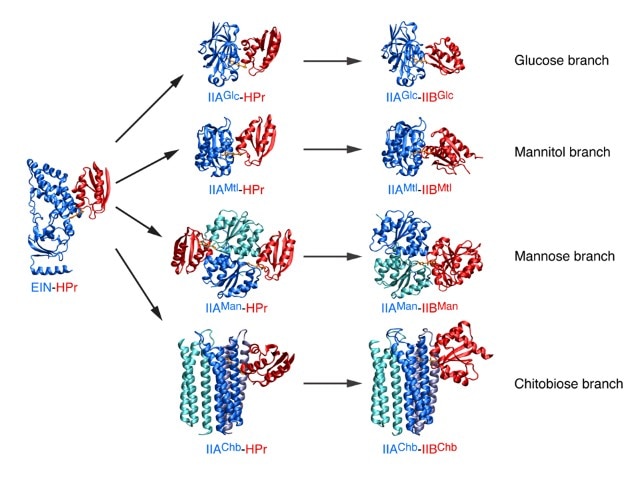
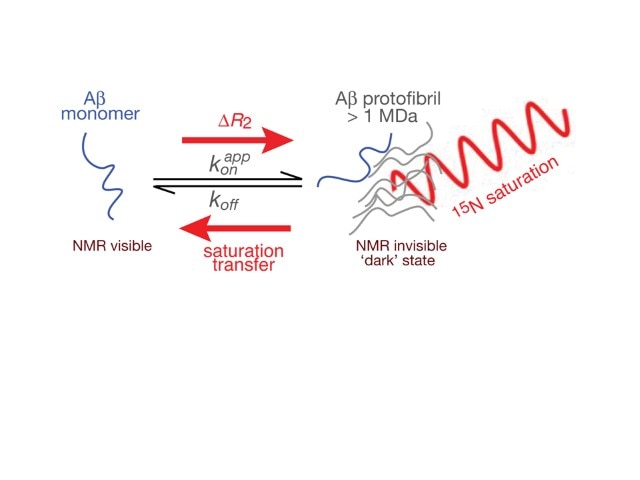
Research Images