Story of Discovery: Progress on the Pathway to Prevention of Type 1 Diabetes
Type 1 diabetes is a devastating illness where the body’s ability to produce the pancreas-derived hormone insulin is lost, requiring people with the disease to administer insulin daily for survival. Even with this burdensome treatment, people with type 1 diabetes are at risk for life-threatening complications. Research shows that the incidence of type 1 diabetes is on the rise in the United States, so identifying ways to prevent type 1 diabetes in those at risk is critical, in parallel with efforts to cure the disease in those who have been diagnosed. The story of type 1 diabetes—which is still being written as key questions and challenges remain— involves geneticists, epidemiologists, molecular and cellular biologists, immunologists, endocrinologists, bioengineers, researchers in other fields, and patient participants. This multifaceted and collaborative approach has resulted in valuable new knowledge that is moving us closer to a long-standing goal of type 1 diabetes prevention.
IDENTIFYING THOSE AT RISK TO DEVELOP TYPE 1 DIABETES
Preventing type 1 diabetes requires not only a successful therapy, but also the ability to identify those who are at risk of developing the disease. But answering the deceptively simple question of “who is at risk?” required a multi-pronged research approach.
One of the first steps was to understand the disease better. Early on, scientists searched for a toxin or infectious agent that caused type 1 diabetes. However, some observed that people with type 1 diabetes sometimes had other disorders related to abnormal hormone levels or function (endocrine disorders), particularly those associated with autoimmunity, leading scientists, after decades of studying the disease, to hypothesize that type 1 diabetes was an autoimmune disease. Autoimmune diseases result when a person’s immune system does not properly distinguish between “self” and “non-self” and inappropriately targets and attacks the body’s own organs, tissues, and cells. One component of an immune attack is antibodies, produced by an immune cell type called B lymphocytes. Self-directed antibodies are called “autoantibodies,” and their presence in the blood can indicate an autoimmune process.
In the early 1970s, researchers found that, by using blood from people with multiple autoimmune endocrine disorders, including type 1 diabetes, they could detect a specific autoantibody response to insulin-producing pancreatic islet tissue. Later research demonstrated that antibodies that react with islet cells could be found in the majority of people with newly diagnosed type 1 diabetes. Further research has identified more than four different autoantibodies specifically enriched in people with type 1 diabetes. One of the earliest autoantibodies to appear, most commonly in younger children, is directed against insulin. Early studies of families with type 1 diabetes in the 1970s led to the observation that the disease often appeared in siblings, indicating that there could be a genetic component to the disease. NIDDK-supported scientists and others soon discovered that human leukocyte antigen (HLA) gene alleles (variant forms of a gene required for the function of another immune cell type—T cells) were associated with type 1 diabetes. With the use of modern tools for genetic analysis, we now know that HLA accounts for approximately 50 percent of the heritability of type 1 diabetes. Additionally, NIDDK-supported researchers and others have since identified more than 50 other genetic loci that contribute to type 1 diabetes susceptibility, accounting for nearly 90 percent of genetic risk in the Caucasian population, which is most affected by type 1 diabetes. Many of these genes are known to be involved in the immune response, further strengthening the understanding of type 1 diabetes as an autoimmune disease. These exciting findings set the stage for efforts to identify those at risk to develop type 1 diabetes and to test immune-modulating therapies to prevent the disease.
A WINDOW OF OPPORTUNITY TO PREVENT TYPE 1 DIABETES
Until the discovery of autoantibodies, it was generally assumed that type 1 diabetes had an acute onset whose first clinical symptoms were the sudden appearance of metabolic abnormalities as a result of the loss of insulin in previously healthy people. Prevention would be difficult in such a disease, as there would be no warning before the clinical appearance of the disease and identifying at-risk individuals would not be possible. Not only did the revelation that type 1 diabetes was an autoimmune disease mean that autoantibodies could possibly be used to identify those at risk before the manifestation of clinical symptoms, but it also suggested that a window of opportunity for prevention might exist. Destruction of the insulin-producing β (beta) cells (which are in the pancreatic islet cell clusters) by an errant immune attack might happen over time, rather than immediately, and perhaps this destruction could be delayed or stopped altogether, preserving the precious remaining β cells.
NIDDK-supported scientists and others spent the 1980s studying cohorts of people that had these autoantibodies in their blood but had not been clinically diagnosed with diabetes to determine whether the appearance of autoantibodies preceded loss of insulin and if they indicated the early stages of type 1 diabetes. In one study, NIDDK-supported investigators followed a set of triplets and a set of twins, each with one person with type 1 diabetes. These people were studied for nearly 2 decades. Over that time, one triplet and one twin—neither of whom had diabetes at the start of the study—first developed autoantibodies and then onset of clinically overt disease, allowing scientists to document the slow, progressive loss of insulin before the onset of clinical diabetes.
In another study, NIDDK-supported scientists screened over 1,700 first-degree relatives (parents, siblings, and offspring) of people with type 1 diabetes for the presence of islet-cell autoantibodies. Only 16 of those screened had the autoantibodies, but 2 of those developed type 1 diabetes in the next 2 years, compared to 1 of the 1,700 without antibodies. In addition, the researchers examined the insulin response in 12 of the relatives with autoantibodies and found that 6 of these individuals had low insulin responses, an indicator of diminished β cell function. Results from these and similar studies contributed to the growing body of evidence that islet-cell autoantibodies were predictors of type 1 diabetes and that β cell destruction was not an immediate event. These studies also provided key information on how screening programs could be designed to identify people and assess their risk, setting the stage for trials to prevent type 1 diabetes.
SETTING THE STAGE FOR A LARGE¬SCALE PREVENTION TRIAL
For a first test of type 1 diabetes prevention, researchers turned to a familiar candidate: insulin. Studies in animal models, as well as small pilot studies in humans, suggested that insulin could delay type 1 diabetes development. It was thought that administering low-dose insulin to an at-risk person before the disease progressed could induce protective immunity that might slow or prevent the immune system’s attack. In 1994, the NIDDK-supported Diabetes Prevention Trial-Type 1 Diabetes (DPT-1) began screening first- and second-degree relatives to identify eligible participants for a clinical trial to test this hypothesis. More than 84,000 people were screened; about 340 were found positive for autoantibodies, had more than an estimated 50 percent chance to develop type 1 diabetes in the next 5 years, and elected to participate in a study testing injectable insulin for prevention of type 1 diabetes. Participants were studied for an average of about 3.5 years, and this clinical trial concluded in 2001. DPT-1 also tested the effect of orally administered insulin in relatives who had an estimated 26 to 50 percent chance of developing type 1 diabetes in the next 5 years. Over 370 participants were studied for an average of 4.3 years in that trial, which concluded in 2003. Although both injectable and oral insulin were very safe, with negligible side effects, neither was found to delay or prevent type 1 diabetes.
Despite the negative results, the DPT-1 was a success in other ways. DPT-1 researchers estimated participant risk using the presence of islet-cell antibodies, insulin response to glucose tests, and the presence or absence of specific HLA alleles, validating these predictive tools and demonstrating that it was possible to identify a cohort of people at high risk for type 1 diabetes. DPT-1 also demonstrated that large type 1 diabetes prevention trials were feasible in at-risk family members of individuals with type 1 diabetes, establishing a path for future prevention trials, just in time for the emergence of new agents that would require testing.
CREATING A NETWORK FOR PREVENTION TRIALS: TYPE 1 DIABETES TRIALNET
As the DPT-1 was concluding, the continued need for a network of investigators and sites to conduct trials of promising therapies to prevent type 1 diabetes became evident. These trials would require screening of large numbers of people to identify those who would be eligible to participate. Additionally, a coordinated and collaborative effort would accelerate progress in this field. Thus, in 2001, NIDDK launched Type 1 Diabetes TrialNet. Since its start nearly 2 decades ago, TrialNet has become an international network of clinical research centers, affiliate sites, a hub, and a coordinating center that involves hundreds of scientists and staff and, most importantly, thousands of participants. TrialNet has conducted multiple studies of agents to delay progression of type 1 diabetes in people with and at risk for the disease, as well as contributed key insights into understanding the type 1 diabetes disease process.
REFINING RISK AND STAGING PROGRESSION OF TYPE 1 DIABETES
The ability to accurately assess those at risk for type 1 diabetes is critical to identify participants for prevention trials and to ensure that as many people as possible can benefit, if and when new prevention strategies are proven effective. To refine and quantify type 1 diabetes risk, NIDDK-supported researchers pooled data from multiple studies and, in 2013, reported that the majority of children who had multiple islet autoantibodies in their blood progressed to the disease over the next 15 years, suggesting that prevention studies focus on this high-risk population.
Data from DPT-1, TrialNet, and other studies revealed that progression to clinical type 1 diabetes proceeds through distinct stages prior to onset of symptoms. This formed the basis for a recommendation from TrialNet, JDRF, the Endocrine Society, and the American Diabetes Association for a type 1 diabetes staging classification in at-risk individuals. This staging provides a framework for the research and development of preventive therapies (see Figure 1): stage 1 is defined as the presence of two or more different types of islet autoantibodies with normal blood glucose (sugar) levels and is considered early type 1 diabetes; stage 2 diabetes is the presence of two or more autoantibodies but with abnormal blood glucose levels without symptoms; and stage 3 is when clinical diagnosis has been reached and symptoms of type 1 diabetes are usually present. TrialNet’s prevention trials enroll individuals with pre-clinical (stage 1 and 2) type 1 diabetes, and TrialNet’s new-onset trials enroll participants in early stage 3 diabetes.
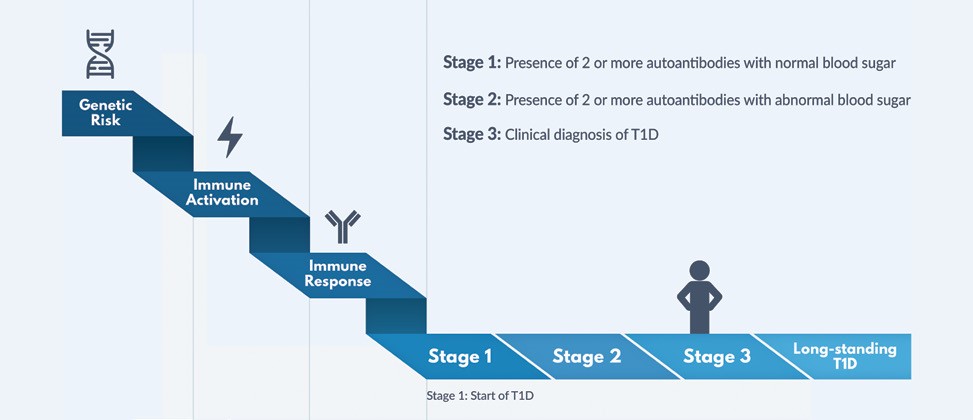 Figure 1: Windows for Prevention of Type 1 Diabetes (T1D): This graphic illustrates how type 1 diabetes progresses. Genetic risk, combined with an unknown environmental trigger(s), is followed by inappropriate activation of the immune system to attack the insulin-producing β cells. The appearance of more than one islet-cell autoantibody in a person’s blood indicates that the immune system has been activated and the person has a high risk of development of type 1 diabetes. Stages 1 and 2 are considered the start of type 1 diabetes, even before the appearance of clinical symptoms and before the clinical diagnosis of diabetes is made—a window of opportunity for strategies to prevent onset of clinical disease. Research by Type 1 Diabetes TrialNet contributed to this new knowledge of type 1 diabetes staging, and TrialNet’s goal is to test agents to prevent or delay the disease at all stages. (Graphic courtesy of Type 1 Diabetes TrialNet.)
Figure 1: Windows for Prevention of Type 1 Diabetes (T1D): This graphic illustrates how type 1 diabetes progresses. Genetic risk, combined with an unknown environmental trigger(s), is followed by inappropriate activation of the immune system to attack the insulin-producing β cells. The appearance of more than one islet-cell autoantibody in a person’s blood indicates that the immune system has been activated and the person has a high risk of development of type 1 diabetes. Stages 1 and 2 are considered the start of type 1 diabetes, even before the appearance of clinical symptoms and before the clinical diagnosis of diabetes is made—a window of opportunity for strategies to prevent onset of clinical disease. Research by Type 1 Diabetes TrialNet contributed to this new knowledge of type 1 diabetes staging, and TrialNet’s goal is to test agents to prevent or delay the disease at all stages. (Graphic courtesy of Type 1 Diabetes TrialNet.)The combination of these efforts led to the following understanding of type 1 diabetes disease risk: 35 percent of people in stage 1 and 70 percent of people in stage 2 will progress to clinical diabetes within 3 to 5 years of identification. The lifetime risk for developing clinical type 1 diabetes from stage 1 or 2 nears 100 percent. In the future, risk assessment could take into account an individual’s genetic makeup and their environmental exposures to determine risk even before autoantibodies appear.
Of note, most new cases of type 1 diabetes occur in people who have no affected relatives. There is currently no way to identify these people other than by conducting population-wide genetic screening, which is not currently feasible. Therefore, for now, research has demonstrated that the most efficient way to identify people at risk for type 1 diabetes is to screen first- and second-degree relatives of people with the disease due to their 15-fold increased risk for developing the disease compared to the general population. To date, TrialNet has screened more than 200,000 relatives and screens more than 15,000 annually to identify at-risk individuals for enrollment in trials. More than 7,000 people have enrolled in TrialNet’s Pathway to Prevention Study (an observational study for relatives with autoantibodies) and/or have participated in a TrialNet trial.
IDENTIFYING CANDIDATE THERAPIES TO TEST
One of the challenges of clinical trials is balancing the potential benefits against the risks. There is risk associated with introducing an agent, particularly one that modulates the immune system and may have serious side effects, into a healthy person— albeit one who will eventually develop clinical diabetes but has not yet done so—especially when the participants are children. Therefore, careful consideration is paramount in deciding which agents are the most promising and should be tested in prevention trials. With that in mind, TrialNet looks for agents that have been tested for safety in animal models, in pilot studies in people, or have been tested (or even approved for use) in people with other autoimmune diseases or conditions before a larger prevention trial is considered.
One of the first agents to emerge as a possible candidate for therapy was an antibody known as anti-CD3, which modifies the function of T cells, but does not dramatically deplete them. In studies of mouse models of type 1 diabetes, anti-CD3 agents have consistently reversed diabetes at the onset of symptoms. In a small clinical trial funded by NIDDK and reported in 2002, scientists found that an anti¬CD3 agent, teplizumab, preserved some insulin production after 1 year with no severe side effects in people recently diagnosed with type 1 diabetes. Following these results, multiple larger and longer studies confirmed that teplizumab treatment delayed the loss of insulin production, including one done by the National Institute of Allergy and Infectious Diseases’ Immune Tolerance Network, an important TrialNet partner.
To continue to make progress towards a more effective, durable and safe therapy for type 1 diabetes, TrialNet has tested additional immune-modifying agents in people newly diagnosed with type 1 diabetes. For example, in 2009, TrialNet reported that rituximab, which is approved as a cancer therapy and destroys B lymphocytes, slowed disease progression for 6 to 9 months. Another TrialNet trial tested the drug abatacept, which acts on T cells and is an approved therapy for rheumatoid arthritis. The results showed that participants who received abatacept had higher insulin production than those who received placebo after 2 years. In 2018, TrialNet reported that another immune system suppressant, low-dose anti-thymocyte globulin (ATG), delayed the loss of insulin production and improved blood glucose control for up to 2 years. All of these drugs can have significant side effects and each had only temporary benefit, likely due in part to immune cell regeneration when the treatments ended. However, these positive results showed that many immune-modulating therapies could slow the disease, indicating that these agents could be tested in prevention trials, where their effects could be more beneficial.
DEMONSTRATION THAT TYPE 1 DIABETES CAN BE PREVENTED
Teplizumab, with several positive trials in people with newly diagnosed type 1 diabetes, was chosen by TrialNet as the first agent to test for disease prevention. TrialNet began a trial in 2011 and, in 2019, reported that teplizumab delayed onset of clinical type 1 diabetes in people at high risk (stage 2) for an average of 2 years (see advance and Patient Profile in this chapter). This exciting discovery provides the first evidence that the onset of clinical type 1 diabetes can be delayed with early preventive treatment. Participants are being followed to determine the durability of the effect, but these results have important implications for people, particularly youth. Treatment with teplizumab could give at-risk individuals 2 years free of type 1 diabetes and insulin administration; 2 years that they do not have to check blood glucose levels; and 2 more years of good health towards preventing or delaying diabetes complications. Based on TrialNet’s results, the U.S. Food and Drug Administration gave teplizumab “Breakthrough Therapy Designation” to expedite its development and review.
THE FUTURE FOR PREVENTION TRIALS OF TYPE 1 DIABETES
Much remains to be explored about teplizumab and other immune-modifying drugs so that more effective treatments can be designed. First and foremost, we need to understand more about the mechanisms of autoimmune pathogenesis and how individual people respond to therapies. From the beginning, TrialNet has engaged in mechanistic research, collecting blood samples from people enrolled in trials and analyzing them for the specific mechanistic effects of treatment. Building on these data, TrialNet has designed a new prevention trial that will combine two agents that showed benefit to newly diagnosed participants in previous trials and that affect complementary immune pathways. Alternative dosing regimens, testing agents even earlier in at-risk people (i.e., stage 1), and other types of combination trials all present exciting opportunities to build on this advance. Additionally, TrialNet currently has two other single-agent prevention studies under way: one testing abatacept (see earlier) and one testing the drug hydroxychloroquine, both of which are already used to reduce symptoms and progression of other autoimmune diseases. There are also many other promising therapies in TrialNet’s pipeline, with even more expected in the future as new knowledge is uncovered by TrialNet’s mechanistic work and through other NIDDK-supported research efforts focused on the underlying mechanisms of type 1 diabetes development. With continued research, the goal of preventing type 1 diabetes—permanently and in anyone who could develop the disease—now seems possible after decades of contributions from countless scientists and, most importantly, the trial participants who never gave up hope.
