Constance Tom Noguchi, Ph.D.

- Acting Chief: Molecular Medicine Branch
- Section Chief: Molecular Cell Biology Section, Molecular Medicine Branch
Professional Experience
- Academic Advisor, FAES (Foundation for Advanced Education in the Sciences, Inc.) Graduate School at the NIH, 2022-2023
- Dean, FAES (Foundation for Advanced Education in the Sciences, Inc.) Graduate School at the NIH, 1999-2022
- Chief, Molecular Cell Biology Section, Molecular Medicine Branch, NIDDK, 1994-present
- Research Physicist, Laboratory of Chemical Biology, NIDDK, 1985-present
- NRSA Fellowship, National Institute of General Medical Sciences, Laboratory of Chemical Biology, NIAMDD, 1975-1977
- Ph.D., the George Washington University, 1975
- B.A., University of California, Berkeley, 1970
Research Goal
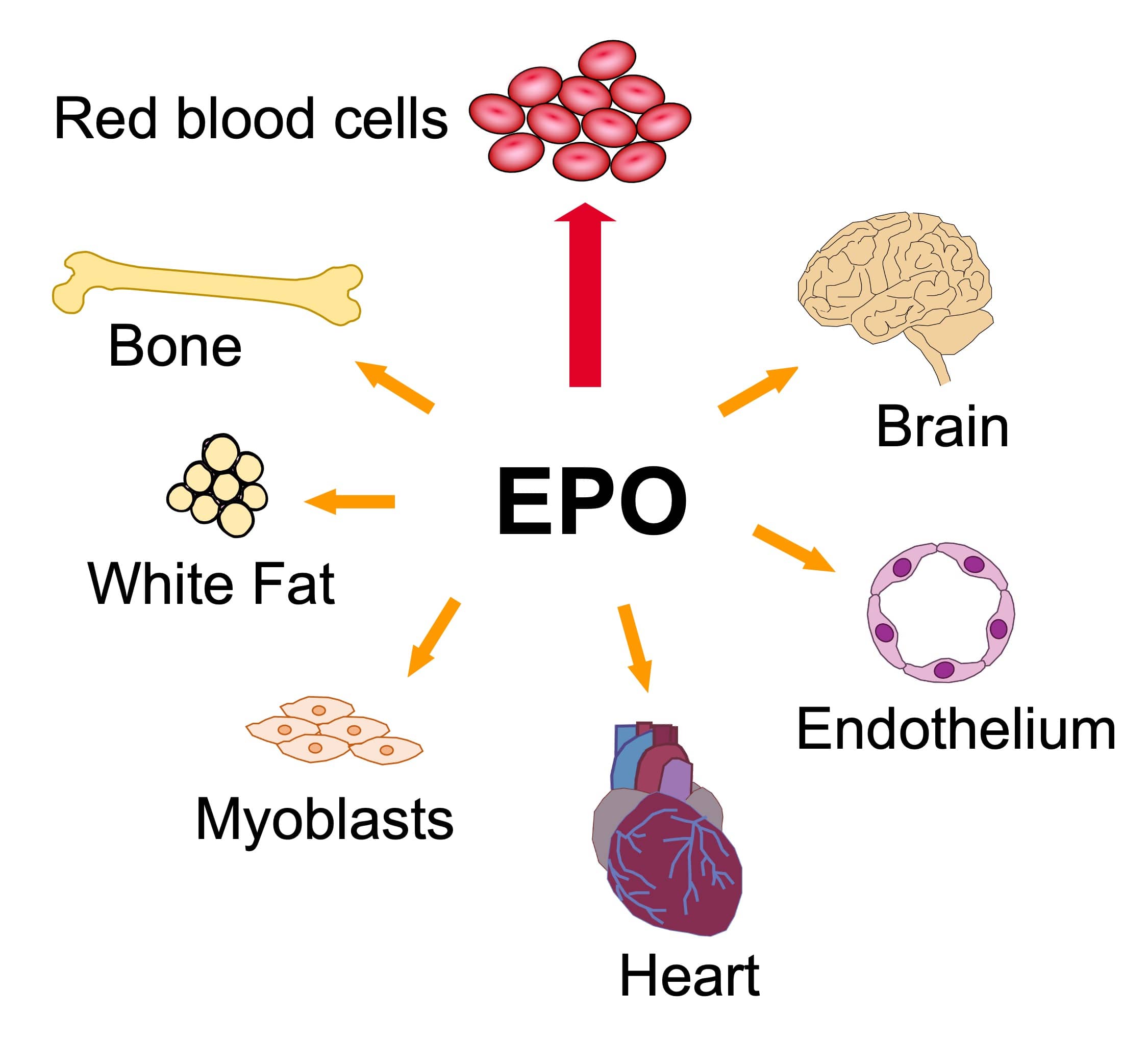
Induction of erythropoietin production by low oxygen enables erythropoietin to act as a protective hormone against hypoxic stress. Elevated erythropoietin stimulates survival, proliferation, and differentiation of blood stem cells/progenitor cells to form mature red blood cells. Erythropoietin activity in other tissues including cardiovascular, brain, muscle, and fat suggests the potential for erythropoietin to act as a pleiotropic hypoxia responsive hormone to facilitate tissue maintenance or repair and wound healing. Erythropoietin stimulation of nitric oxide production in vascular endothelium contributes to increased oxygen delivery. Erythropoietin activity in non-erythroid tissue complements its function in erythroid and endothelial tissue to increase oxygen delivery by promoting oxygen consumption.
The purpose of these studies on erythropoietin biology is to determine endogenous erythropoietin activity in erythroid and non-erythroid tissue, including response to hypoxia, and the action of erythropoietin treatment to regulate red blood cell production, oxygen delivery, and other tissue responses.
Current Research
Our lab investigates the role of erythropoietin in maintaining stem/progenitor cell characteristics. Molecular structure and processing related to cellular function, differentiation, and development are studied using molecular and cell biology, biochemical and biophysical approaches. Emphasis is on transcriptional regulation of proliferation and differentiation in animal models. We are particularly interested in the function of the erythropoietin receptor and metabolic and stress response to erythropoietin in hematopoietic, neuronal, muscle, endothelial, bone, gender-specific regulation of fat mass and other stem/progenitor cells. Other studies include erythroid differentiation, globin gene expression and related pathophysiology of sickle cell disease and other hemoglobinopathies.
Applying our Research
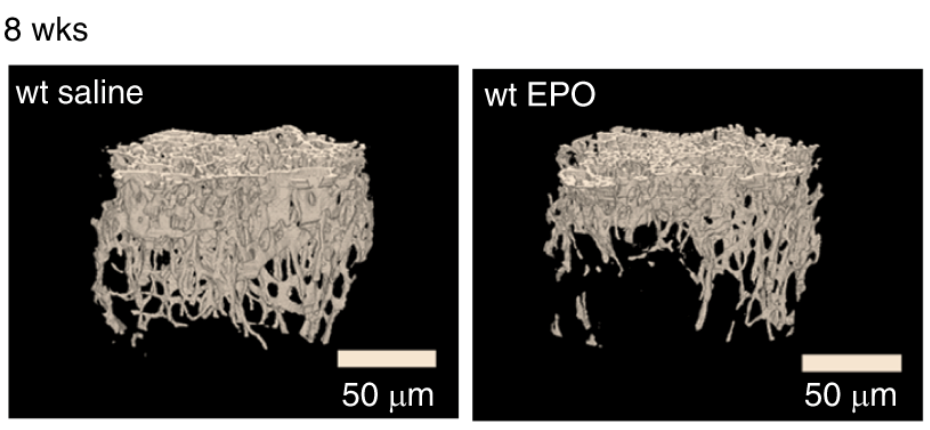
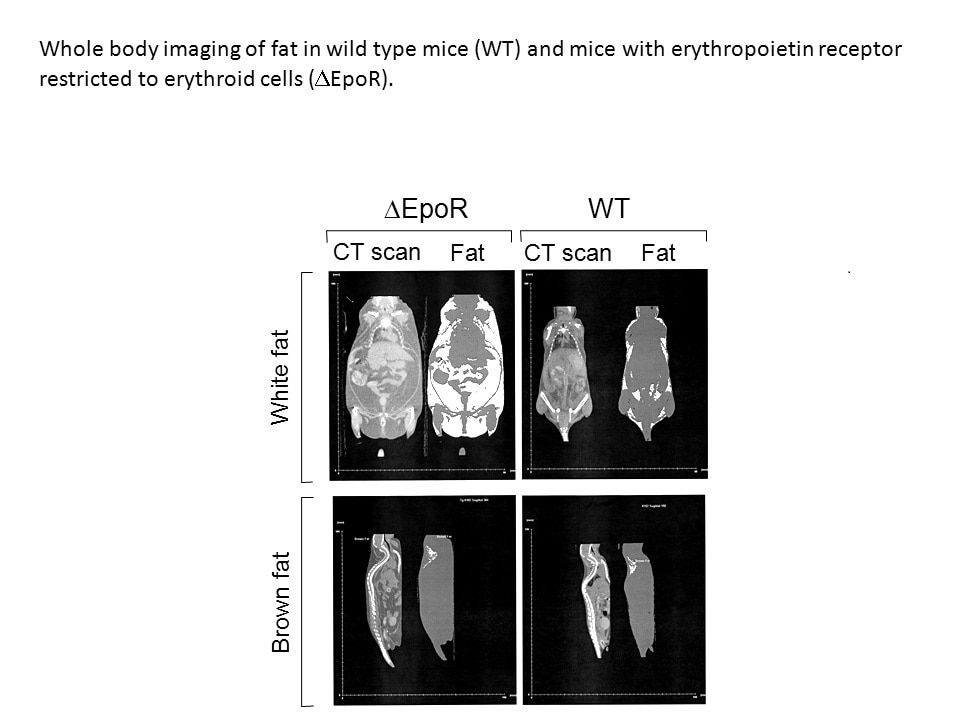
Erythropoietin has been available as a therapeutic treatment to increase red blood cell production for treatment of anemia. We have found that during diet-induced obesity in mice, erythropoietin regulation of metabolism decreases fat mass accumulation with associated inflammation, an effect that is gender-specific, evident in males and abrogated by estrogen in females. Increased brain erythropoietin decreases fat mass and obesity associated inflammation in the hypothalamus in male and ovariectomized female mice. Erythropoietin also regulates bone marrow adipogenesis. In mice, erythropoietin treatment reduces bone marrow adipocytes before and independent of change in white adipose tissue mass and promotes bone loss in male and female mice independent of erythropoietin stimulated red blood cell production.
Efforts to broaden clinical applications of erythropoietin, including high dose erythropoietin treatment in anemic patients to increase hematocrit to normal levels in chronic kidney disease and select cancers, led to awareness of adverse effects associated with high dose treatment. It also caused the Food and Drug Administration (FDA) to reduce the approved dosage for erythropoietin therapy. Animal models suggest that erythropoietin dose may also adversely affect bone health. Multivariable analysis of the United States Renal Data System (USRDS) datasets for 1997-2013 revealed an association of erythropoietin dose as an independent risk factor for hip fracture in dialysis patients.
Full characterization of endogenous erythropoietin activity and response to erythropoietin administration in tissues beyond red blood cell production will provide insight on its use to treat anemia and associated risks and possible related adverse events. It will also provide the potential application of erythropoietin administration for stroke, cardiovascular, metabolic and other diseases, or tissue injury.
Need for Further Study
Erythropoietin activity in animal models and cultured cells has been useful in identifying erythropoietin action in erythroid and various other tissues. Areas for further study include endothelial and cardiovascular response to erythropoietin affecting oxygen delivery and promoting tissue repair. Erythropoietin stimulation of nitric oxide production suggests a potential link between nitric oxide synthase and erythropoietin in erythropoietic response and other tissue specific activity. Erythropoietin response in non-erythroid tissues such as white fat and brain contributes to regulation of metabolism, glucose tolerance and fat mass in white adipose tissue, especially in obese male mice. Erythropoietin regulation of fat mass and glucose metabolism in mice that become more obese with age suggests that erythropoietin metabolic response may be age/sex dependent and that erythropoietin stimulation may affect other fat depots. Other non-hematopoietic tissues may contribute to erythropoietin metabolic regulation including the gut and intestinal microbiota. Erythropoietin stimulated lipid metabolism during erythropoiesis and lipid uptake in non-hematopoietic tissue requires clarification.
Understanding these aspects of erythropoietin action in addition to the response of blood cells and regulation of red blood cell production will clarify the role of stem cell/progenitor cell and tissue specific response to erythropoietin during development and tissue maintenance and repair. Potential non-hematopoietic erythropoietin activity may give rise to potential beneficial or adverse response accompanying erythropoietin treatment for anemia and for specific tissue injury or disease.
Select Publications
- Erythropoietin regulates energy metabolism through EPO-EpoR-RUNX1 axis.
- Yin W, Rajvanshi PK, Rogers HM, Yoshida T, Kopp JB, An X, Gassmann M, Noguchi CT.
- Nat Commun (2024 Sep 16) 15:8114. Abstract/Full Text
- Erythropoietin treatment and the risk of hip fractures in hemodialysis patients.
- Suresh S, Wright EC, Wright DG, Abbott KC, Noguchi CT.
- J Bone Miner Res (2021 Jul) 36:1211-1219. Abstract/Full Text
Research in Plain Language
Stem cells and progenitor cells have the remarkable ability to develop into different cell types. They offer the potential to provide a renewable source of replacement cells to treat diseases, conditions, and disabilities. We use molecular and cell biology, biochemical, and biophysical approaches to learn how these cells work. We are also particularly interested in the role of cytokines and hormones that trigger the initial development of stem and progenitor cells into different cell types. Our research allows us to build models of how these processes work.
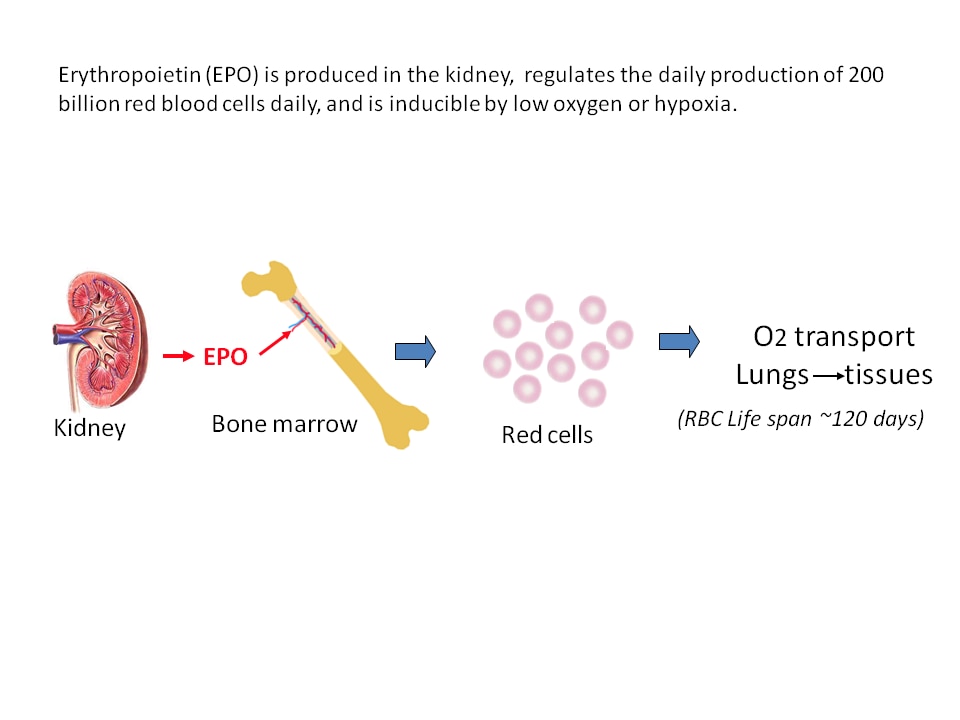
One focus of our lab’s research is on the way in which a stem cell produces a red blood cell offspring. Erythropoietin, produced in the kidney, is the hormone that regulates the daily production of 200 billion new red blood cells. These red blood cells are important because they carry oxygen from the lungs to tissues. In our lab we investigate many roles of erythropoietin such as the following:
- When oxygen levels are low, erythropoietin is triggered to increase red blood cell formation. As more red blood cells are formed, more oxygen can be moved from the lungs to tissues. This increases overall oxygen levels in the body.
- Erythropoietin stimulates endothelial cells to produce increased nitric oxide production; this regulates blood flow and contributes to increasing oxygen delivery. We relate this activity to a protective effect in a mouse model of heart ischemic injury (blocked blood flow).
- Erythropoietin can stimulate muscle progenitor cells to promote wound healing in a mouse model of skeletal muscle injury.
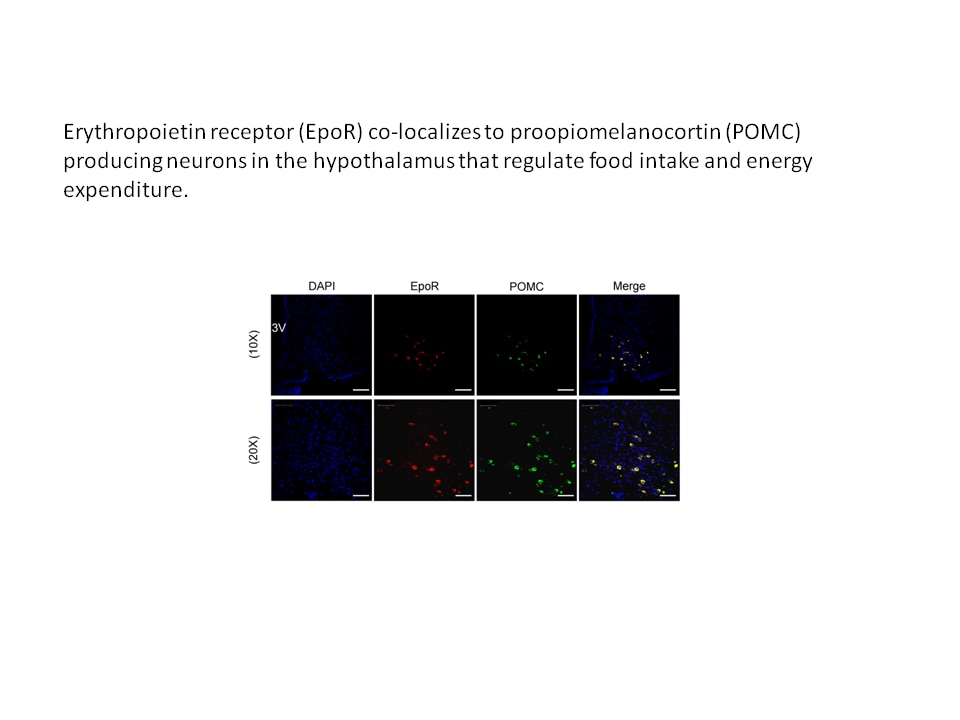
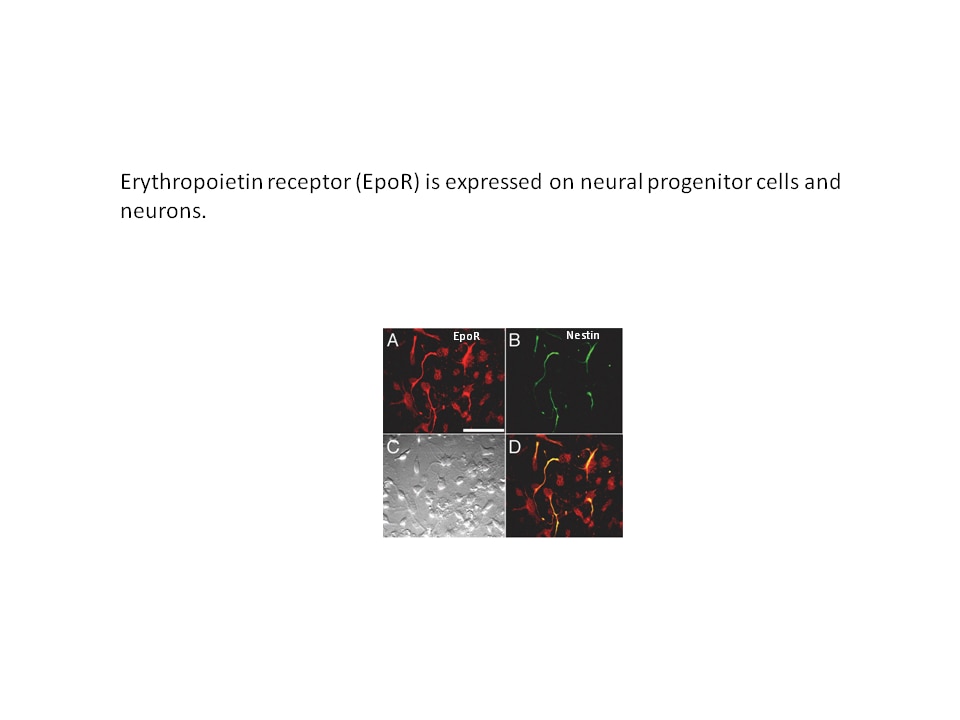
- In the brain, erythropoietin can stimulate the proliferation of neural progenitor cells and the survival of neurons to provide protection in mouse models of ischemic injury. Erythropoietin activity in brain also contributes to improved glucose metabolism and protection against diet-induced obesity.
- Erythropoietin contributes to fat tissue metabolic response. Erythropoietin treatment, especially in obese mice, improves glucose metabolism and insulin sensitivity, increases energy expenditure and activity, and in male mice reduces food intake and fat mass. Female mice exhibit erythropoietin regulation of fat mass after disruption of the anti-obesity effect of estrogen.
- Erythropoietin stimulates bone forming cells in the bone marrow to regulate bone and bone marrow fat, an activity required for normal bone formation in mice. Erythropoietin treatment to increase red blood cell production is accompanied by decrease in bone marrow fat and a direct response of bone forming progenitor cells resulting in bone loss.
Other studies in our lab focus on the activity of the globin gene family. Globin genes are components of hemoglobin, the molecule that carries oxygen. These genes make up 98 percent of the protein in red blood cells that transport oxygen in the bloodstream. In particular, we want to understand the changes in hemoglobin function observed in sickle cell disease and other diseases related to hemoglobin abnormalities.
Research Images