Yoshitasu Sei, M.D., Ph.D.
Professional Experience
- Staff Scientist, NIDDK, NIH, 2009-Present
- Laboratory Head, GCPP, NIMH, 2004-2009
- Assistant Professor, USUHS, 1996-2004
- Visiting Associate, NIDDK, 1991-1996
- Visiting Fellow, NIDDK, 1988-1991
- Visiting Scientist, NY Mount Sinai Medical Center, 1985-1988
- PhD,Kurume University School of Medicine, Japan, 1988
- MD, Kurume University School of Medicine, Japan, 1983
Research Goal
Our goal is to understand cellular and molecular biological mechanisms of differentiation and tumorigenesis of enteroendocrine cells.
Current Research
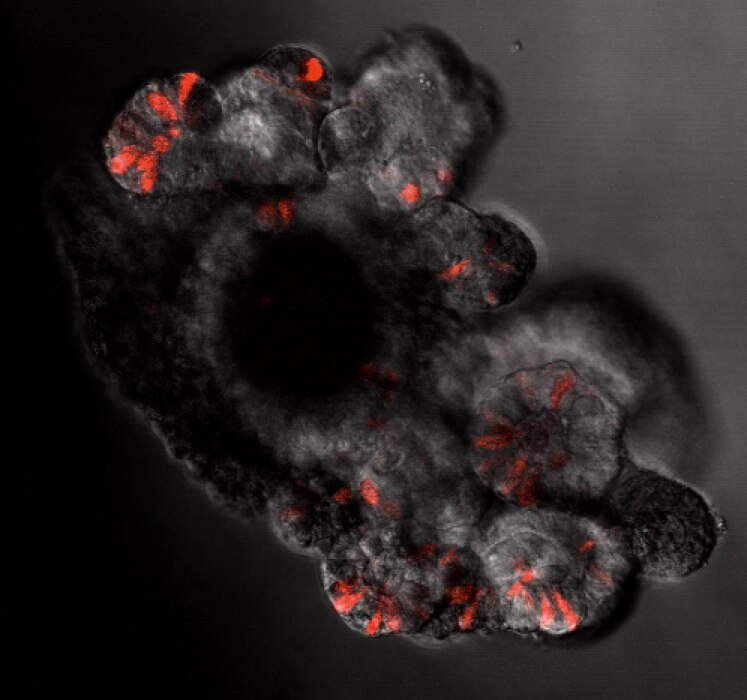
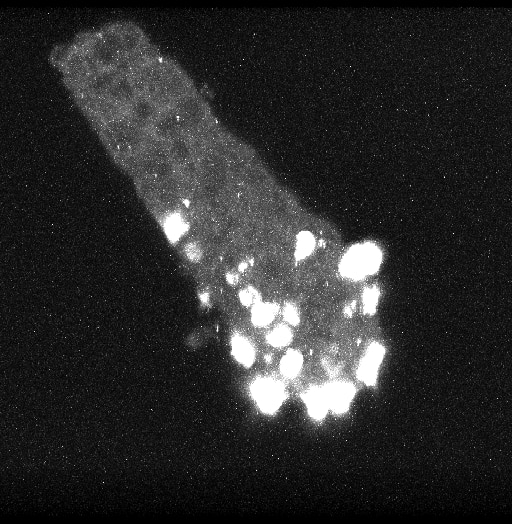
Small intestinal neuroendocrine tumors (SI-NET) (previously termed midgut carcinoid tumors) are serotonin-secreting well differentiated neuroendocrine tumors of putative enterochromaffin (EC) cell origin. Despite recent genome-wide genetic studies, basic mechanisms underlying initiation and progression of SI-NETS are unclear. Our approach to study tumorigenesis of SI-NETs has been progressed by integrating clinically-oriented studies of familial SI-NETs and basic research on molecular and cell biological mechanisms of enteroendocrine differentiation. Our recent studies recognize a subset of EC cells that is label-retaining at the +4 position in the crypt and functions as a reserve intestinal stem cell. Importantly, this +4 reserve EC cell subset not only contributes to regeneration of the intestinal epithelium during injury and inflammation but also to basal crypt homeostasis at a constant rate. The latter function suggests that the +4 EC cell subset serves as an active reserve stem cell via a constant rate of de-differentiation. Characterization of early tumor formation of SI-NET, observed as crypt-based EC cell clusters in many cases of familial SI-NETs, suggests that the +4 active reserve EC cell subset is the cell of origin. This newly discovered active reserve stem cell property of EC cells can account for unique biological mechanisms and processes associated with the genesis and development of SI-NETs. The recognition of this property of the +4 active reserve EC cell subset may provide novel opportunities to explore NETs in the gastrointestinal tract as well other organs. We use lineage-tracing, organoid cultures, whole transcriptome analysis, FACS-cell sorting and single cell analysis to accelerate our understanding of +4 active reserve EC cell subset.
Applying our Research
The research on endocrine cells in the intestine has been subjected to potential new drug discoveries and therapeutics for diseases widely affecting public health including obesity, diabetes, and digestive diseases such as irritable bowel syndrome and neuroendocrine tumors.
Need for Further Study
Further studies are needed to understand how cell molecular signaling and chromosome remodeling cooperate to regulate cell fate and differentiation.
Select Publications
- Diagnostic value of whole-mount crypt analysis of ileal biopsy specimens for the patients with familial small intestinal neuroendocrine tumors.
- Sei Y, Forbes J, Da B, Chitsaz E, Feng J, Zhao X, Hughes MS, Wank SA.
- Ther Adv Med Oncol (2023) 15:17588359231156871. Abstract/Full Text
- Tissue- and cell-specific properties of enterochromaffin cells affect the fate of tumorigenesis toward nonendocrine adenocarcinoma of the small intestine.
- Sei Y, Feng J, Zhao X, Dagur P, McCoy JP, Merchant JL, Wank SA.
- Am J Physiol Gastrointest Liver Physiol (2023 Mar 1) 324:G177-G189. Abstract/Full Text
Research in Plain Language
Our research focuses on studying hormone-producing cells in the intestine. Hormone-producing cells (enteroendocrine cells) produce hormones by sensing chemicals used to regulate physiological functions in the digestive system. We use genetically engineered mouse models and a gut model in the culture dish to study how the enteroendocrine cells differentiate from intestinal stem cells to mature hormone producing cells. Recently, we discovered a group of enteroendocrine cells that express stem cell markers and stay in the stem cell area. We think this group of enteroendocrine cells may have a unique capability in terms of stemness in certain conditions and that this group of cells may be responsible for the generation of enteroendocrine tumors, including carcinoids. Studying this group of cells may help scientists discover how endocrine tumors are generated and therefore help them create new drugs to treat these tumors.
Research Images