A New Artificial Pancreas System for People with Type 1 Diabetes
NIH-funded study shows new system better controls blood glucose than current technology.
Results from a multicenter randomized clinical trial evaluating a new artificial pancreas system — which automatically monitors and regulates blood glucose levels — found that the new system was more effective than existing treatments at controlling blood glucose levels in people with type 1 diabetes.
The International Diabetes Closed-Loop (iDCL) Study showed that the system improved participants’ blood glucose control throughout the day and overnight. The latter is a common but serious challenge for children and adults with type 1 diabetes, since blood glucose can drop to dangerously low levels when a person is asleep.
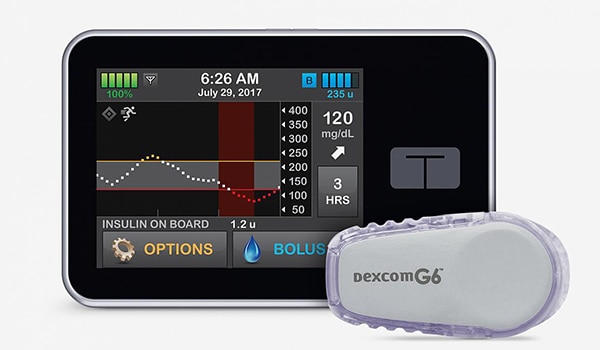
The artificial pancreas, also known as closed-loop control, is an “all-in-one” diabetes management system that tracks blood glucose levels using a continuous glucose monitor (CGM) and automatically delivers the hormone insulin when needed using an insulin pump. The system could replace depending on fingerstick tests or CGM with separate delivery of insulin by multiple daily injections or a pump.
“Artificial pancreas technology has tremendous potential to improve the day-to-day lives of people with type 1 diabetes,” said NIDDK Director Griffin P. Rodgers, MD. “By making management of type 1 diabetes easier and more precise, this technology could reduce the daily burden of this disease, while also potentially reducing diabetes complications including eye, nerve, and kidney diseases.”
The iDCL Study is one of four major research efforts funded by NIDDK through the Special Statutory Funding Program for Type 1 Diabetes to test and refine advanced artificial pancreas systems. This six-month study using the new system was the third phase in a series of five iDCL artificial pancreas clinical protocols implemented by 10 research centers in the United States and Europe. The study was conducted with participants living their usual day-to-day lives, so the researchers could best understand how the system works in typical daily routines.
The research is published in the New England Journal of Medicine and results from this study have been submitted to the U.S. Food and Drug Administration for approval to market the system. For more information, view the full NIH press release.
Note: Since this post was published, the FDA has approved the marketing of the Tandem Diabetes Care Control-IQ Technology that was featured in the International Diabetes Closed-Loop (iDCL) Study.
