Susanne Neumann, Ph.D.

- Senior Associate Scientist: Laboratory of Endocrinology & Receptor Biology
- Senior Associate Scientist (On Detail): Office of Scientific Director
Professional Experience
- Staff Scientist, Laboratory of Endocrinology and Receptor Biology, NIDDK, NIH, 2006–Present
- Postdoctoral Fellow, Clinical Endocrinology Branch, NIDDK, NIH, 2002–2006
- Postdoctoral Fellow, University of Leipzig, III. Medical Department/Endocrinology and Diabetes Branch, 2000–2001
- Ph.D., University of Leipzig, III. Medical Department/Endocrinology and Diabetes Branch, 2000
- Member of the American Thyroid Association since 2005
Research Goal
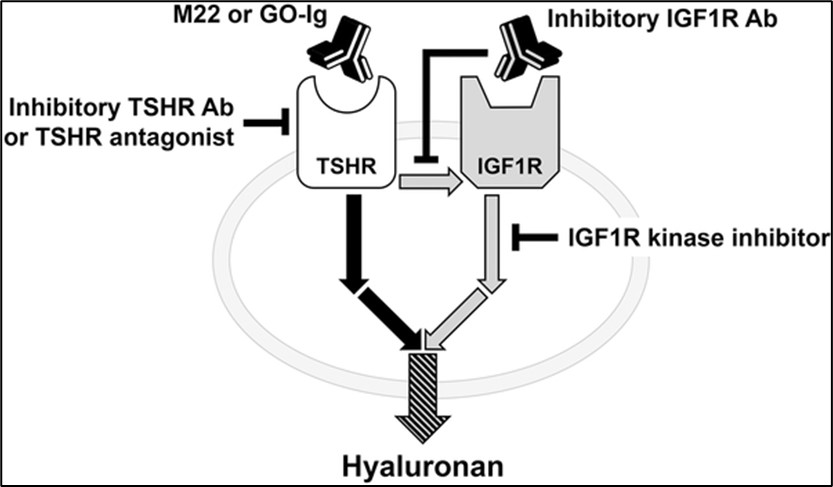
We work on the development of orally available thyrotropin receptor (TSHR) agonists for the diagnosis and treatment of thyroid cancer. A major focus of our research program is also the development of TSHR antagonists that might have potential for the treatment of Graves' hyperthyroidism, an autoimmune disease, and associated Thyroid Eye Disease (TED). Our group studies also the molecular mechanisms involved in the pathogenesis of Graves' hyperthyroidism and TED with focus on crosstalk between the TSHR and the IGF1-receptor (IGF1R) and their downstream signaling cascades.
Current Research
TSH activates TSH receptors (TSHR), thereby stimulating the function of thyroid follicular cells (thyrocytes). This leads to the biosynthesis and secretion of thyroid hormones. Several thyroid pathologies are associated with TSHR, providing a strong argument for the design of small molecule ligands (SML). Our focus is the development of SMLs that agonize or antagonize the TSHR.
rhTSH (Thyrogen®, Genzyme) has been used in the follow-up of patients with thyroid cancer to increase the sensitivity for detection of recurrent or metastatic disease. rhTSH, which is a heterodimeric 30 kilodalton glycoprotein, is difficult to produce and must be administered by injection. A small molecule TSHR agonist could produce the same beneficial effects as rhTSH but with greater ease of production and the benefit of oral administration. We developed a SML that is a full agonist at TSHR and has clinical potential. We are working on the further development of this agonist and its test in preclinical studies. Moreover, we use this agonist as a probe to study the molecular mechanism of TSHR activation and function in cells in tissue culture and in animal models.
Graves’ hyperthyroidism (GH) is caused by persistent, unregulated stimulation of thyroid cells by thyroid-stimulating antibodies (TSAbs) that activate the TSHR. A small molecule antagonist that directly targets the TSHR could have therapeutic potential for GH patients. We identified the first small molecule TSHR antagonists and demonstrated their effects in primary cultures of human thyrocytes and in an animal model. Another possible clinical application of an antagonist beyond treatment of Graves' hyperthyroidism could be to alleviate the symptoms of TED. Therefore, we extended our studies to primary cultures of human orbital fibroblasts which are obtained from TED patients who have undergone orbital decompression surgery. Our recent findings suggest that drug-like TSHR antagonists may have clinical potential for the treatment of TED, and might allow to treat patients with GH and TED with the same drug. We are working on the further development of our TSHR antagonist lead compounds to be able to conduct studies in animal models of TED with the goal of subsequent preclinical tests.
Applying our Research
Patients with thyroid cancer could benefit from the development of a drug-like small molecule TSHR agonist. An orally available small molecule TSHR agonist will be available to a much larger patient population worldwide than human recombinant TSH that needs to be injected. Patients with Graves' hyperthyroidism and Thyroid Eye Disease could benefit from the development of a small molecule TSHR antagonist.
Need for Further Study
Our results provide proof-of-principle for effectiveness of small molecule agonists and antagonists for TSHR. Chemical optimization of these small molecule ligands needs to be performed in an effort to produce more potent molecules for testing in animal models and for future clinical development.
Furthermore, these molecules can now be used as probes to study TSHR physiology and pathophysiology in cell culture and in animal models. Crosstalk between TSHR and IGF1R plays a role in the pathophysiology of Thyroid Eye Disease. Small molecule antagonists for the TSHR allow us to study the underlying molecular mechanisms of TSHR/IGF1R crosstalk and to develop a potential treatment for Thyroid Eye Disease.
Select Publications
- Inhibition of TSH Receptor Expression by a Cyclotriazadisulfonamide as a Potential Treatment of Graves Hyperthyroidism.
- Krieger CC, Neumann S, Sui X, Templin JS, Kapri T, Demillo VG, Olsen RK, Intasiri A, Gershengorn MC, Bell TW.
- Endocrinology (2025 Feb 27) 166. Abstract/Full Text
- Linsitinib Decreases Thyrotropin-Induced Thyroid Hormone Synthesis by Inhibiting Crosstalk Between Thyroid-Stimulating Hormone and Insulin-Like Growth Factor 1 Receptors in Human Thyrocytes In Vitro and In Vivo in Mice.
- Boutin A, Eliseeva E, Templin S, Marcus-Samuels B, Anderson DE, Gershengorn MC, Neumann S.
- Thyroid (2025 Feb) 35:216-224. Abstract/Full Text
Research in Plain Language
The thyroid is a butterfly-shaped gland in the neck. The TSH receptor (TSHR) is expressed in the thyroid and stimulated by the hormone TSH. The TSHR stimulates the function of thyroid follicular cells, the thyrocytes. This leads to biosynthesis (the production of chemical compounds) and the secretion of thyroid hormones.
Several thyroid pathogens (infectious agents that cause disease) are associated with TSHR. This provides a strong argument for the design of drug-like organic small molecule ligands (SMLs) that will influence TSHR action. Our focus is the development of SMLs that agonize (stimulate) or antagonize (inhibit) TSHR action.
A TSHR agonist will be useful for patients with thyroid cancer. Iodide uptake by thyroid cells is the first step in thyroid hormone synthesis and is very important for the sensitivity of the diagnosis of thyroid cancer. A TSHR agonist stimulates uptake of iodide by thyroid cancer cells, and therefore, can help to diagnose residual cancer tissue.
On the other hand, there are patients with an autoimmune disease, Graves’ hyperthyroidism (GH). Antibodies in the serum of these patients permanently stimulate the TSHR, leading to an unregulated stimulation of thyroid cells. A small molecule antagonist that inhibits the continuous stimulation of the TSHR by antibodies could be useful in the therapy of GH and Thyroid Eye Disease (TED), a disease that is closely associated with GH.
We have provided proof-of-principle for the effectiveness of small molecule agonists and antagonists for TSHR and are now focusing on the further development of these molecules. We are preparing them for testing in animal models and for future clinical development.
Related Links
- Kai Ge, Laboratory of Endocrinology & Receptor Biology
- Douglas Forrest, Nuclear Receptor Biology Section
Research Images