Research Updates
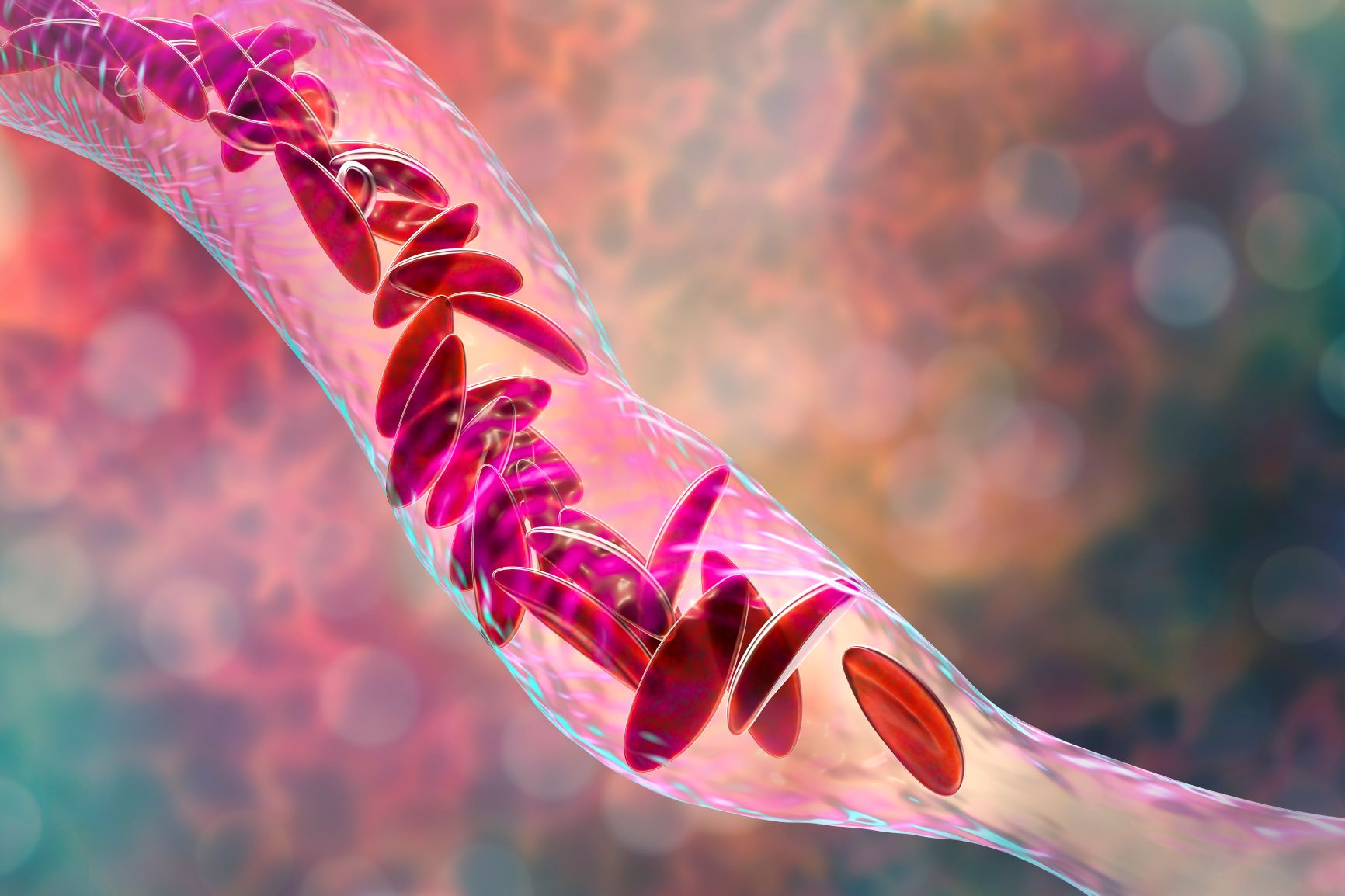
NIH study finds encouraging results for gene therapy for severe sickle cell disease: Early results from a human clinical trial testing a novel gene replacement therapy in people with severe sickle cell disease suggest that the approach has an acceptable level of safety and might help patients consistently produce normal red blood cells instead of the sickle-shaped ones that mark this painful, life-threatening disease. The experimental treatment involves removing hematopoietic stem cells from the person’s bone marrow or blood and adding a therapeutic beta globin gene, which is defective in people with sickle cell disease. The cells are then returned to the person, leading to the production of anti-sickling hemoglobin (T87Q). Based on these preliminary findings, researchers believe the new gene replacement therapy will enable the person’s bone marrow to produce normal red blood cells consistently. This research was published in December in Blood.
Mega docking library poised to speed drug discovery: Researchers have launched an ultra-large virtual docking library expected to grow to more than 1 billion molecules by next year. The library enables unbiased computational modeling of various chemical compounds, vastly expediting the process of discovering new treatments by allowing researchers to virtually “see” a molecule docking with its receptor protein and predict its pharmacological properties, based on how the molecular structures are predicted to interact. Only those relatively few candidate molecules that best match the target profile on the computer need to be physically made and tested in a wet lab. The larger the library, the better its odds of weeding out inactive “decoy” molecules that could otherwise lead researchers down blind alleys. The research, funded by the NIH’s Common Fund Illuminating the Druggable Genome (IDG) Program, was published in Nature in February.
Major NIH-funded clinical trial adds nuanced evidence linking brain and vascular health: Intensive lowering of blood pressure did not significantly reduce dementia risk but did have a measurable impact on mild cognitive impairment (MCI), according to new results from the Systolic Blood Pressure Intervention Trial Memory and Cognition in Decreased Hypertension (SPRINT MIND). SPRINT MIND secondary results are the first to show an intervention that significantly reduces the occurrence of MCI, which is a well-established precursor of dementia. The results were reported in the Journal of the American Medical Association in January. SPRINT MIND was an integral aspect of the initial design for SPRINT, a large, randomized NIH-funded clinical trial looking at the effects of intensive blood pressure lowering on cardiovascular and renal disease.
Researchers find genetic vulnerability to menthol cigarette use: A genetic variant found only in people of African descent significantly increases a smoker’s preference for cigarettes containing menthol, a flavor additive. The variant of the MRGPRX4 gene is five to eight times more frequent among smokers who use menthol cigarettes than other smokers, according to an international group of researchers supported by NIH and the U.S. Food and Drug Administration. The multiethnic study is the first to look across all genes to identify genetic vulnerability to menthol cigarettes. The paper was published online in the journal PLOS Genetics in February.
Patient stem cell-derived adipocytes reveal genetic variation that predicts antidiabetic drug response: Thiazolidinedione drugs, or TZDs, are one of the most potent classes of drugs for reversing insulin resistance in type 2 diabetes. However, risks of serious cardiovascular complications have significantly reduced their use, and notably, 20-30 percent of people taking TZDs fail to respond to the drug’s therapeutic effects. A recent NIH-funded study evaluated genetic variations among people with obesity and how these differences determine which people will respond to the drug and which people will not. The study also showed that a single mutation near a gene involved in cholesterol metabolism, ABCA1, determined whether those taking TZDs would experience an increase blood cholesterol levels, a known side-effect that is linked to heart attack and stroke. The research published in Cell Stem Cell in December.

‘Exercise hormone’ tied to bone-strengthening benefits: A recent NIH-funded study has shed light on an important mechanism behind the bone-strengthening benefit of exercise. The hormone irisin, discovered in 2012, is secreted by muscles during exercise, and has been shown to mediate beneficial effects on adipose tissues, the brain, and bone. Researchers identified for the first time the receptor for irisin, the alpha-V class of integrins found on the surface of osteocytes, or bone cells. This binding promotes survival of osteocytes and activates secretion of the protein sclerosin, which plays a critical role in bone remodeling. These results, published in December in Cell, suggest that irisin could be a target for the treatment of osteoporosis, which effects approximately 53 million people in the United States.
Elevated hormone flags liver problems in mice with methylmalonic acidemia: NIH researchers discovered that a hormone, fibroblast growth factor 21 (FGF21), is extremely elevated in mice with liver disease that mimics the same condition in patients with methylmalonic acidemia (MMA). MMA is a genomic disease that impairs a person’s ability to break down food proteins and certain fatty acids. Based on the discovery, clinicians treating people with MMA will be able to measure FGF21 levels to predict how severely their livers are affected and when to refer them for liver transplants. The findings also might shed light on more common disorders such as fatty liver disease, obesity, and diabetes by uncovering similarities in how MMA and these disorders affect energy metabolism. This research was published in JCI Insight in December 2018.
Probiotics not helpful for young children with diarrhea: An NIH-supported trial recently found that there is no benefit to treating children experiencing acute gastroenteritis with probiotics. The study included 971 children, ages 3 months to 4 years, who had visited 10 emergency departments across the United States for treatment of acute gastroenteritis, marked by diarrhea, cramping in the abdomen, nausea, and vomiting. Half of the children received a five-day course of the probiotic Lactobacillus rhamnosus GG, and the other half received a placebo. After a two-week period of follow up, the researchers observed no significant differences among the groups regarding severity of symptoms, frequency or duration of diarrhea or vomiting, rate of household transmission, or duration of day care or work absenteeism. The study published in November in the New England Journal of Medicine.
Healthy infants harbor intestinal bacteria that protect against food allergy: Intestinal microbes play a critical role in regulating allergic responses, according to a study published in January in Nature Medicine. Researchers transplanted gut microbes from infant donors with and without milk allergy into groups of mice with allergic antibodies to milk. When later exposed to milk, mice receiving microbes from milk-allergic children produced allergic antibodies and experienced anaphylaxis, a potentially life-threatening allergic reaction. Mice receiving gut microbes from non-allergic infants had no reactions. Further experiments identified that the microbe, Anaerostipes caccae, prevented the development of milk allergy when transplanted alone into groups of mice. These findings suggest that further research could lead to microbiome-modifying therapies to prevent or treat food allergy.

