Thyroid Disease & Pregnancy
On this page:
- What role do thyroid hormones play in pregnancy?
- Hyperthyroidism in Pregnancy
- Hypothyroidism in Pregnancy
- Postpartum Thyroiditis
- Is it safe to breastfeed while I’m taking beta-blockers, thyroid hormone, or antithyroid medicines?
- Thyroid Disease and Eating During Pregnancy
- Clinical Trials
Thyroid disease is a group of disorders that affects the thyroid gland. The thyroid is a small, butterfly-shaped gland in the front of your neck that makes thyroid hormones. Thyroid hormones control how your body uses energy, so they affect the way nearly every organ in your body works—even the way your heart beats.
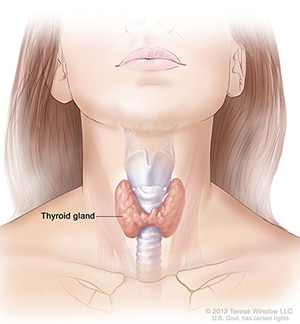
Sometimes the thyroid makes too much or too little of these hormones. Too much thyroid hormone is called hyperthyroidism and can cause many of your body’s functions to speed up. “Hyper” means the thyroid is overactive. Learn more about hyperthyroidism in pregnancy. Too little thyroid hormone is called hypothyroidism and can cause many of your body’s functions to slow down. “Hypo” means the thyroid is underactive. Learn more about hypothyroidism in pregnancy.
If you have thyroid problems, you can still have a healthy pregnancy and protect your baby’s health by having regular thyroid function tests and taking any medicines that your doctor prescribes.
What role do thyroid hormones play in pregnancy?
Thyroid hormones are crucial for normal development of your baby’s brain and nervous system. During the first trimester—the first 3 months of pregnancy—your baby depends on your supply of thyroid hormone, which comes through the placenta. At around 12 weeks, your baby’s thyroid starts to work on its own, but it doesn’t make enough thyroid hormone until 18 to 20 weeks of pregnancy.
Two pregnancy-related hormones—human chorionic gonadotropin (hCG) and estrogen—cause higher measured thyroid hormone levels in your blood. The thyroid enlarges slightly in healthy women during pregnancy, but usually not enough for a health care professional to feel during a physical exam.
Thyroid problems can be hard to diagnose in pregnancy due to higher levels of thyroid hormones and other symptoms that occur in both pregnancy and thyroid disorders. Some symptoms of hyperthyroidism or hypothyroidism are easier to spot and may prompt your doctor to test you for these thyroid diseases.
Another type of thyroid disease, postpartum thyroiditis, can occur after your baby is born.
Hyperthyroidism in Pregnancy
What are the symptoms of hyperthyroidism in pregnancy?
Some signs and symptoms of hyperthyroidism often occur in normal pregnancies, including faster heart rate, trouble dealing with heat, and tiredness.
Other signs and symptoms can suggest hyperthyroidism:
- fast and irregular heartbeat
- shaky hands
- unexplained weight loss or failure to have normal pregnancy weight gain
What causes hyperthyroidism in pregnancy?
Hyperthyroidism in pregnancy is usually caused by Graves’ disease and occurs in 1 to 4 of every 1,000 pregnancies in the United States.1 Graves’ disease is an autoimmune disorder. With this disease, your immune system makes antibodies that cause the thyroid to make too much thyroid hormone. This antibody is called thyroid stimulating immunoglobulin, or TSI.
Graves’ disease may first appear during pregnancy. However, if you already have Graves’ disease, your symptoms could improve in your second and third trimesters. Some parts of your immune system are less active later in pregnancy so your immune system makes less TSI. This may be why symptoms improve. Graves’ disease often gets worse again in the first few months after your baby is born, when TSI levels go up again. If you have Graves’ disease, your doctor will most likely test your thyroid function monthly throughout your pregnancy and may need to treat your hyperthyroidism.1 Thyroid hormone levels that are too high can harm your health and your baby’s.
Rarely, hyperthyroidism in pregnancy is linked to hyperemesis gravidarum—severe nausea and vomiting that can lead to weight loss and dehydration. Experts believe this severe nausea and vomiting is caused by high levels of hCG early in pregnancy. High hCG levels can cause the thyroid to make too much thyroid hormone. This type of hyperthyroidism usually goes away during the second half of pregnancy.
Less often, one or more nodules, or lumps in your thyroid, make too much thyroid hormone.
How can hyperthyroidism affect me and my baby?
Untreated hyperthyroidism during pregnancy can lead to
- miscarriage
- premature birth
- low birthweight
- preeclampsia—a dangerous rise in blood pressure in late pregnancy
- thyroid storm—a sudden, severe worsening of symptoms
- congestive heart failure
Rarely, Graves’ disease may also affect a baby’s thyroid, causing it to make too much thyroid hormone. Even if your hyperthyroidism was cured by radioactive iodine treatment to destroy thyroid cells or surgery to remove your thyroid, your body still makes the TSI antibody. When levels of this antibody are high, TSI may travel to your baby’s bloodstream. Just as TSI caused your own thyroid to make too much thyroid hormone, it can also cause your baby’s thyroid to make too much.
Tell your doctor if you’ve had surgery or radioactive iodine treatment for Graves’ disease so he or she can check your TSI levels. If they are very high, your doctor will monitor your baby for thyroid-related problems later in your pregnancy.

An overactive thyroid in a newborn can lead to
- a fast heart rate, which can lead to heart failure
- early closing of the soft spot in the baby’s skull
- poor weight gain
- irritability
Sometimes an enlarged thyroid can press against your baby’s windpipe and make it hard for your baby to breathe. If you have Graves’ disease, your health care team should closely monitor you and your newborn.
How do doctors diagnose hyperthyroidism in pregnancy?
Your doctor will review your symptoms and do some blood tests to measure your thyroid hormone levels. Your doctor may also look for antibodies in your blood to see if Graves’ disease is causing your hyperthyroidism. Learn more about thyroid tests and what the results mean.
How do doctors treat hyperthyroidism during pregnancy?
If you have mild hyperthyroidism during pregnancy, you probably won’t need treatment. If your hyperthyroidism is linked to hyperemesis gravidarum, you only need treatment for vomiting and dehydration.
If your hyperthyroidism is more severe, your doctor may prescribe antithyroid medicines, which cause your thyroid to make less thyroid hormone. This treatment prevents too much of your thyroid hormone from getting into your baby’s bloodstream. You may want to see a specialist, such as an endocrinologist or expert in maternal-fetal medicine, who can carefully monitor your baby to make sure you’re getting the right dose.
Doctors most often treat pregnant women with the antithyroid medicine propylthiouracil (PTU) during the first 3 months of pregnancy. Another type of antithyroid medicine, methimazole, is easier to take and has fewer side effects, but is slightly more likely to cause serious birth defects than PTU. Birth defects with either type of medicine are rare. Sometimes doctors switch to methimazole after the first trimester of pregnancy. Some women no longer need antithyroid medicine in the third trimester.
Small amounts of antithyroid medicine move into the baby’s bloodstream and lower the amount of thyroid hormone the baby makes. If you take antithyroid medicine, your doctor will prescribe the lowest possible dose to avoid hypothyroidism in your baby but enough to treat the high thyroid hormone levels that can also affect your baby.
Antithyroid medicines can cause side effects in some people, including
- allergic reactions such as rashes and itching
- rarely, a decrease in the number of white blood cells in the body, which can make it harder for your body to fight infection
- liver failure, in rare cases
Stop your antithyroid medicine and call your doctor right away if you develop any of these symptoms while taking antithyroid medicines:
- yellowing of your skin or the whites of your eyes, called jaundice
- dull pain in your abdomen
- constant sore throat
- fever
If you don’t hear back from your doctor the same day, you should go to the nearest emergency room.
You should also contact your doctor if any of these symptoms develop for the first time while you’re taking antithyroid medicines:
- increased tiredness or weakness
- loss of appetite
- skin rash or itching
- easy bruising
If you are allergic to or have severe side effects from antithyroid medicines, your doctor may consider surgery to remove part or most of your thyroid gland. The best time for thyroid surgery during pregnancy is in the second trimester.
Radioactive iodine treatment is not an option for pregnant women because it can damage the baby’s thyroid gland.
Hypothyroidism in Pregnancy
What are the symptoms of hypothyroidism in pregnancy?
Symptoms of an underactive thyroid are often the same for pregnant women as for other people with hypothyroidism. Symptoms include
- extreme tiredness
- trouble dealing with cold
- muscle cramps
- severe constipation
- problems with memory or concentration

Most cases of hypothyroidism in pregnancy are mild and may not have symptoms.
What causes hypothyroidism in pregnancy?
Hypothyroidism in pregnancy is usually caused by Hashimoto’s disease and occurs in 2 to 3 out of every 100 pregnancies.1 Hashimoto’s disease is an autoimmune disorder. In Hashimoto’s disease, the immune system makes antibodies that attack the thyroid, causing inflammation and damage that make it less able to make thyroid hormones.
How can hypothyroidism affect me and my baby?
Untreated hypothyroidism during pregnancy can lead to
- preeclampsia—a dangerous rise in blood pressure in late pregnancy
- anemia
- miscarriage
- low birthweight
- stillbirth
- congestive heart failure, rarely
These problems occur most often with severe hypothyroidism.
Because thyroid hormones are so important to your baby’s brain and nervous system development, untreated hypothyroidism—especially during the first trimester—can cause low IQ and problems with normal development.
How do doctors diagnose hypothyroidism in pregnancy?
Your doctor will review your symptoms and do some blood tests to measure your thyroid hormone levels. Your doctor may also look for certain antibodies in your blood to see if Hashimoto’s disease is causing your hypothyroidism. Learn more about thyroid tests and what the results mean.
How do doctors treat hypothyroidism during pregnancy?
Treatment for hypothyroidism involves replacing the hormone that your own thyroid can no longer make. Your doctor will most likely prescribe levothyroxine, a thyroid hormone medicine that is the same as T4, one of the hormones the thyroid normally makes. Levothyroxine is safe for your baby and especially important until your baby can make his or her own thyroid hormone.
Your thyroid makes a second type of hormone, T3. Early in pregnancy, T3 can’t enter your baby’s brain like T4 can. Instead, any T3 that your baby’s brain needs is made from T4. T3 is included in a lot of thyroid medicines made with animal thyroid, such as Armour Thyroid, but is not useful for your baby’s brain development. These medicines contain too much T3 and not enough T4, and should not be used during pregnancy. Experts recommend only using levothyroxine (T4) while you’re pregnant.
Some women with subclinical hypothyroidism—a mild form of the disease with no clear symptoms—may not need treatment.

If you had hypothyroidism before you became pregnant and are taking levothyroxine, you will probably need to increase your dose. Most thyroid specialists recommend taking two extra doses of thyroid medicine per week, starting right away. Contact your doctor as soon as you know you’re pregnant.
Your doctor will most likely test your thyroid hormone levels every 4 to 6 weeks for the first half of your pregnancy, and at least once after 30 weeks.1 You may need to adjust your dose a few times.
Postpartum Thyroiditis
What is postpartum thyroiditis?
Postpartum thyroiditis is an inflammation of the thyroid that affects about 1 in 20 women during the first year after giving birth1 and is more common in women with type 1 diabetes. The inflammation causes stored thyroid hormone to leak out of your thyroid gland. At first, the leakage raises the hormone levels in your blood, leading to hyperthyroidism. The hyperthyroidism may last up to 3 months. After that, some damage to your thyroid may cause it to become underactive. Your hypothyroidism may last up to a year after your baby is born. However, in some women, hypothyroidism doesn’t go away.
Not all women who have postpartum thyroiditis go through both phases. Some only go through the hyperthyroid phase, and some only the hypothyroid phase.
What are the symptoms of postpartum thyroiditis?
The hyperthyroid phase often has no symptoms—or only mild ones. Symptoms may include irritability, trouble dealing with heat, tiredness, trouble sleeping, and fast heartbeat.
Symptoms of the hypothyroid phase may be mistaken for the “baby blues”—the tiredness and moodiness that sometimes occur after the baby is born. Symptoms of hypothyroidism may also include trouble dealing with cold; dry skin; trouble concentrating; and tingling in your hands, arms, feet, or legs. If these symptoms occur in the first few months after your baby is born or you develop postpartum depression, talk with your doctor as soon as possible.
What causes postpartum thyroiditis?
Postpartum thyroiditis is an autoimmune condition similar to Hashimoto’s disease. If you have postpartum thyroiditis, you may have already had a mild form of autoimmune thyroiditis that flares up after you give birth.

How do doctors diagnose postpartum thyroiditis?
If you have symptoms of postpartum thyroiditis, your doctor will order blood tests to check your thyroid hormone levels.
How do doctors treat postpartum thyroiditis?
The hyperthyroid stage of postpartum thyroiditis rarely needs treatment. If your symptoms are bothering you, your doctor may prescribe a beta-blocker, a medicine that slows your heart rate. Antithyroid medicines are not useful in postpartum thyroiditis, but if you have Grave’s disease, it may worsen after your baby is born and you may need antithyroid medicines.
You’re more likely to have symptoms during the hypothyroid stage. Your doctor may prescribe thyroid hormone medicine to help with your symptoms. If your hypothyroidism doesn’t go away, you will need to take thyroid hormone medicine for the rest of your life.
Is it safe to breastfeed while I’m taking beta-blockers, thyroid hormone, or antithyroid medicines?
Certain beta-blockers are safe to use while you’re breastfeeding because only a small amount shows up in breast milk. The lowest possible dose to relieve your symptoms is best. Only a small amount of thyroid hormone medicine reaches your baby through breast milk, so it’s safe to take while you’re breastfeeding. However, in the case of antithyroid drugs, your doctor will most likely limit your dose to no more than 20 milligrams (mg) of methimazole or, less commonly, 400 mg of PTU.
Thyroid Disease and Eating During Pregnancy
What should I eat during pregnancy to help keep my thyroid and my baby’s thyroid working well?
Because the thyroid uses iodine to make thyroid hormone, iodine is an important mineral for you while you’re pregnant. During pregnancy, your baby gets iodine from your diet. You’ll need more iodine when you’re pregnant—about 250 micrograms a day.1 Good sources of iodine are dairy foods, seafood, eggs, meat, poultry, and iodized salt—salt with added iodine. Experts recommend taking a prenatal vitamin with 150 micrograms of iodine to make sure you’re getting enough, especially if you don’t use iodized salt.1 You also need more iodine while you’re breastfeeding since your baby gets iodine from breast milk. However, too much iodine from supplements such as seaweed can cause thyroid problems. Talk with your doctor about an eating plan that’s right for you and what supplements you should take. Learn more about a healthy diet and nutrition during pregnancy.
Clinical Trials
The National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) and other components of the National Institutes of Health (NIH) conduct and support research into many diseases and conditions.
What are clinical trials, and are they right for you?
Clinical trials are part of clinical research and at the heart of all medical advances. Clinical trials look at new ways to prevent, detect, or treat disease. Researchers also use clinical trials to look at other aspects of care, such as improving the quality of life for people with chronic illnesses. Find out if clinical trials are right for you.
What clinical trials are open?
Clinical trials that are currently open and are recruiting can be viewed at ClinicalTrials.gov.
References
This content is provided as a service of the National Institute of Diabetes and Digestive and Kidney Diseases
(NIDDK), part of the National Institutes of Health. NIDDK translates and disseminates research findings to increase knowledge and understanding about health and disease among patients, health professionals, and the public. Content produced by NIDDK is carefully reviewed by NIDDK scientists and other experts.
The NIDDK would like to thank:
Linda Barbour, M.D., M.S.P.H., FACP, University of Colorado School of Medicine
