About Our Research
The Immunology Section performs basic and translational research to understand (1) innate and adaptive immune responses to hepatitis viruses and (2) the regulation of the hepatic and systemic immune responses by the microbiome.
Current Research
Immunopathogenesis of viral hepatitis
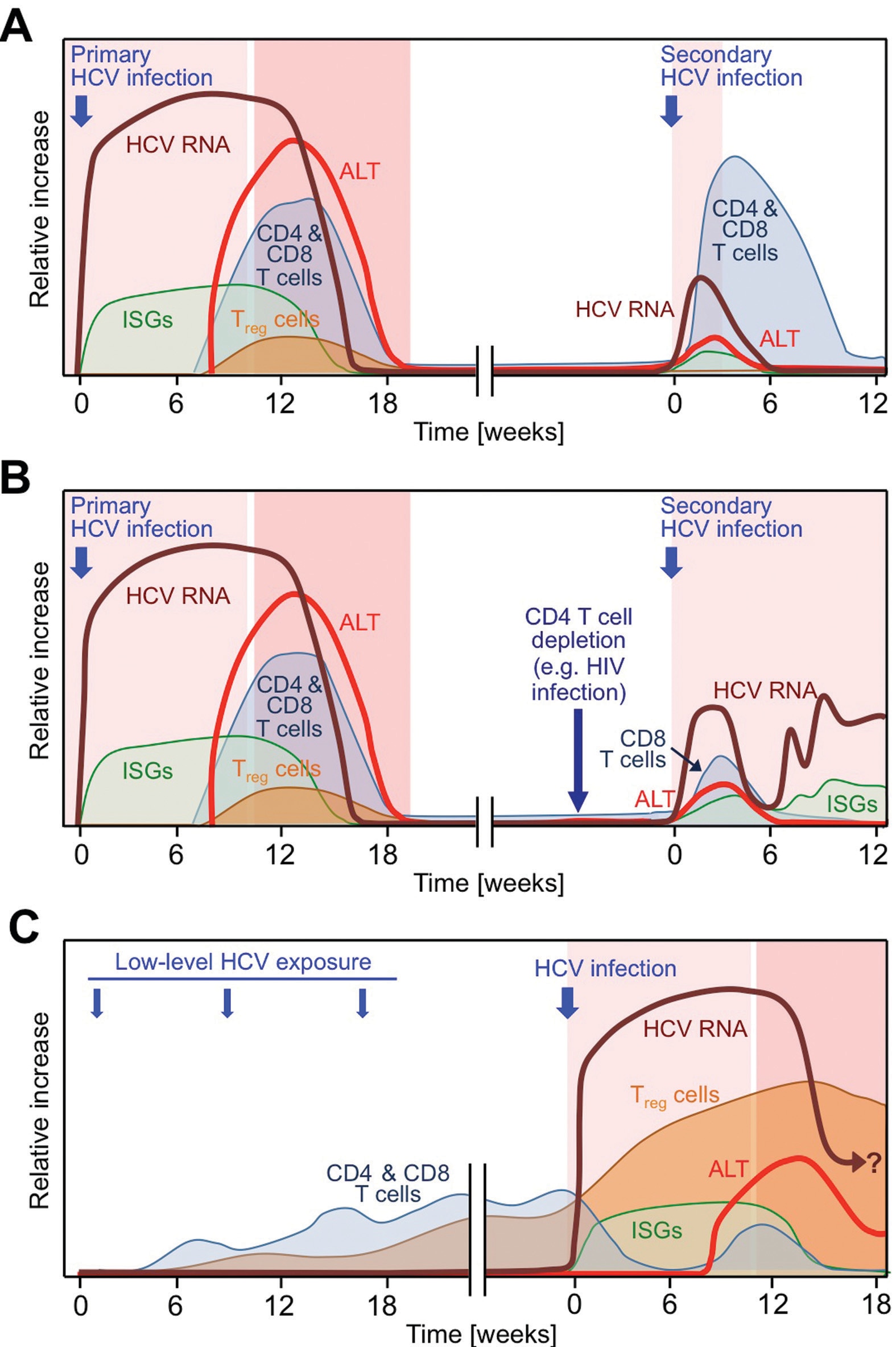
Worldwide mortality from chronic hepatitis is increasing and by 2040 is projected to exceed the combined mortality of HIV infection, tuberculosis and malaria. Three human hepatitis viruses (HBV, HCV and HDV) are the causative agents for almost all cases of viral hepatitis.
View full-sized image
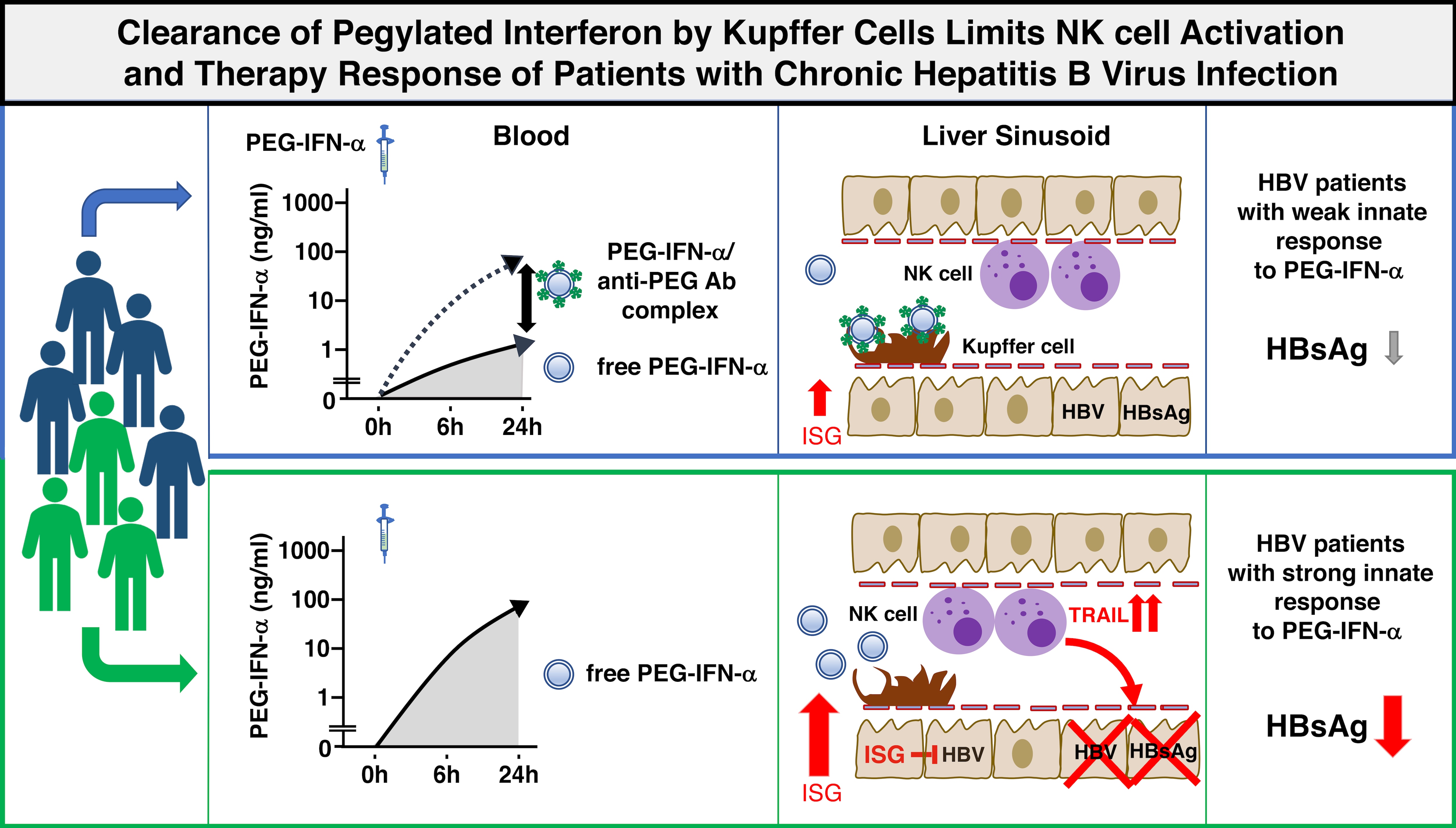
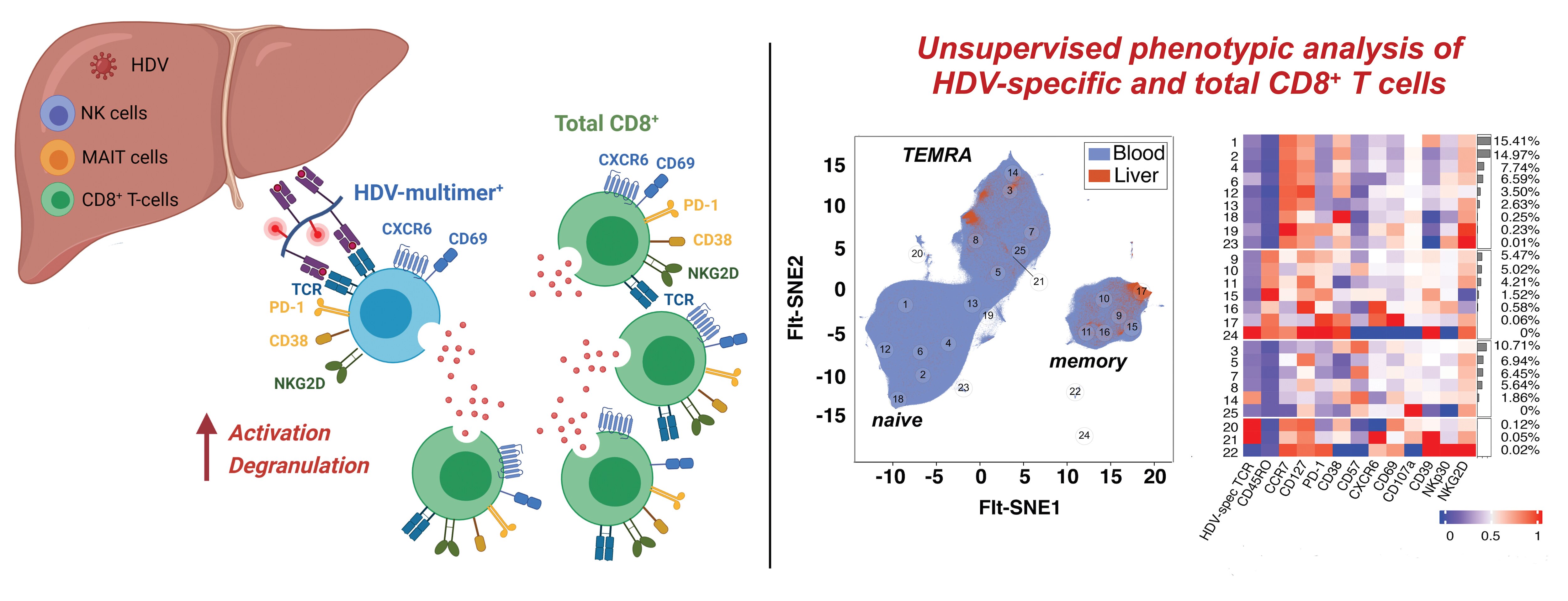
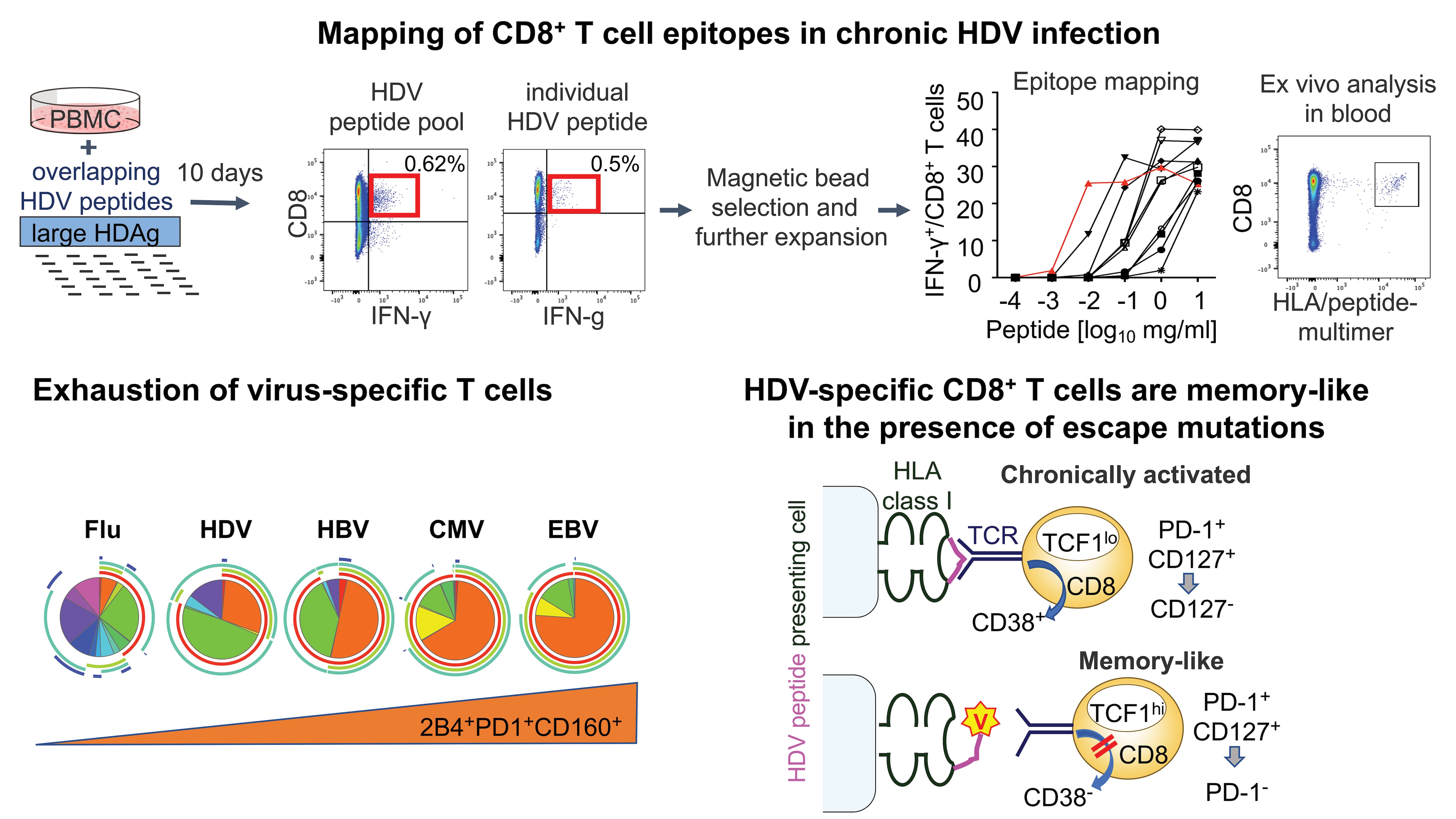
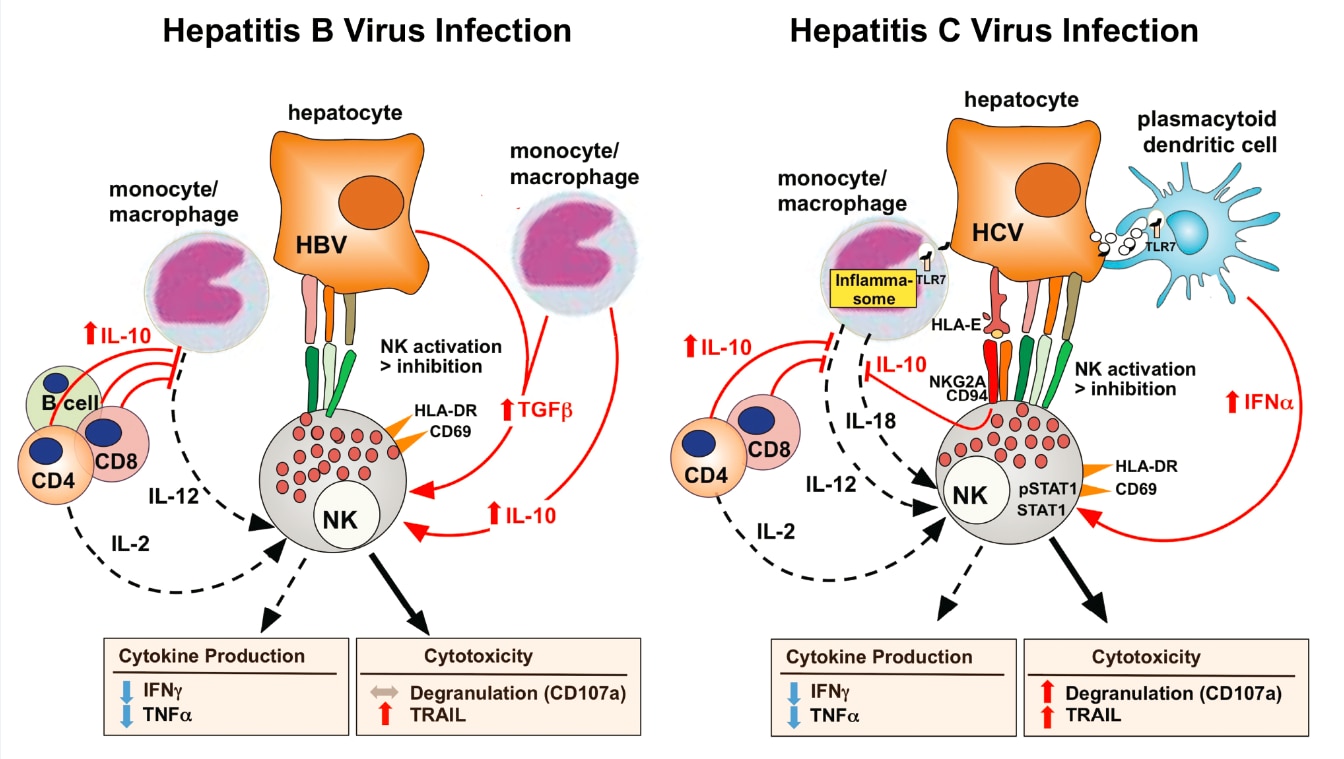
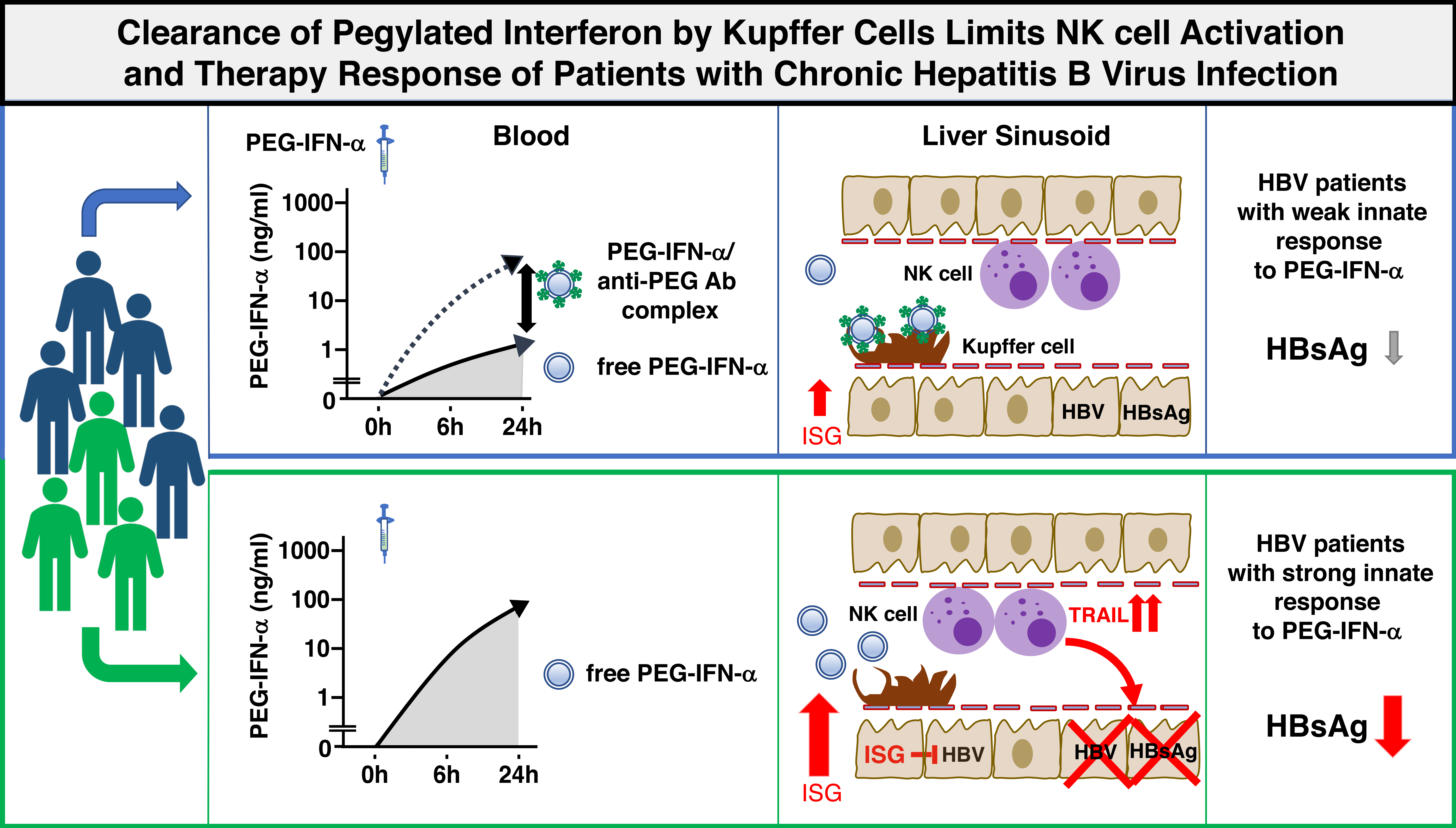 HBV is responsible for 240 million chronic infections worldwide, with a substantial proportion due to vertical transmission from mother to child. HBV is a small DNA virus that establishes a mini chromosome within the hepatocyte nucleus as its transcriptional template and integrates randomly into the host genome. Therefore, HBV cannot be eliminated with antiviral therapy. HDV is an enveloped, closed circular RNA virus that requires HBV to propagate. HDV/HBV co-infection rapidly progresses to cirrhosis in most patients, and there is no effective therapy. HCV is a positive-stranded RNA virus responsible for about 170 million chronic infections worldwide. Highly effective antiviral therapies against HCV are now available, but a preventive HCV vaccine is still needed for worldwide elimination of HCV.
HBV is responsible for 240 million chronic infections worldwide, with a substantial proportion due to vertical transmission from mother to child. HBV is a small DNA virus that establishes a mini chromosome within the hepatocyte nucleus as its transcriptional template and integrates randomly into the host genome. Therefore, HBV cannot be eliminated with antiviral therapy. HDV is an enveloped, closed circular RNA virus that requires HBV to propagate. HDV/HBV co-infection rapidly progresses to cirrhosis in most patients, and there is no effective therapy. HCV is a positive-stranded RNA virus responsible for about 170 million chronic infections worldwide. Highly effective antiviral therapies against HCV are now available, but a preventive HCV vaccine is still needed for worldwide elimination of HCV.
We currently take the following approaches:
- Develop immunological biomarkers and immunomodulatory therapies for a cure of chronic HBV. Specifically, we are studying immune responses in proof-of concept treatment studies of chronic HBV infection (see Nishio et al., Science Translational Medicine 2021)
- Determine the nature and induction of protective immune responses against HCV, which is essential for the development of a prophylactic vaccine. In addition, we are studying biosamples from cured patients to evaluate how innate and adaptive immune responses change when virus and inflammation are removed after decades of infection (see Nishio et al., Nature Communications 2022).
Regulation of hepatic and systemic immune responses by the gut microbiome
The microbiota of mammals co-evolved with their respective hosts over time resulting in a symbiotic relationship. However, the microbiota of humans also exhibits increasing changes to external effects of the modern world and disease-related changes in individuals.
View full-sized image
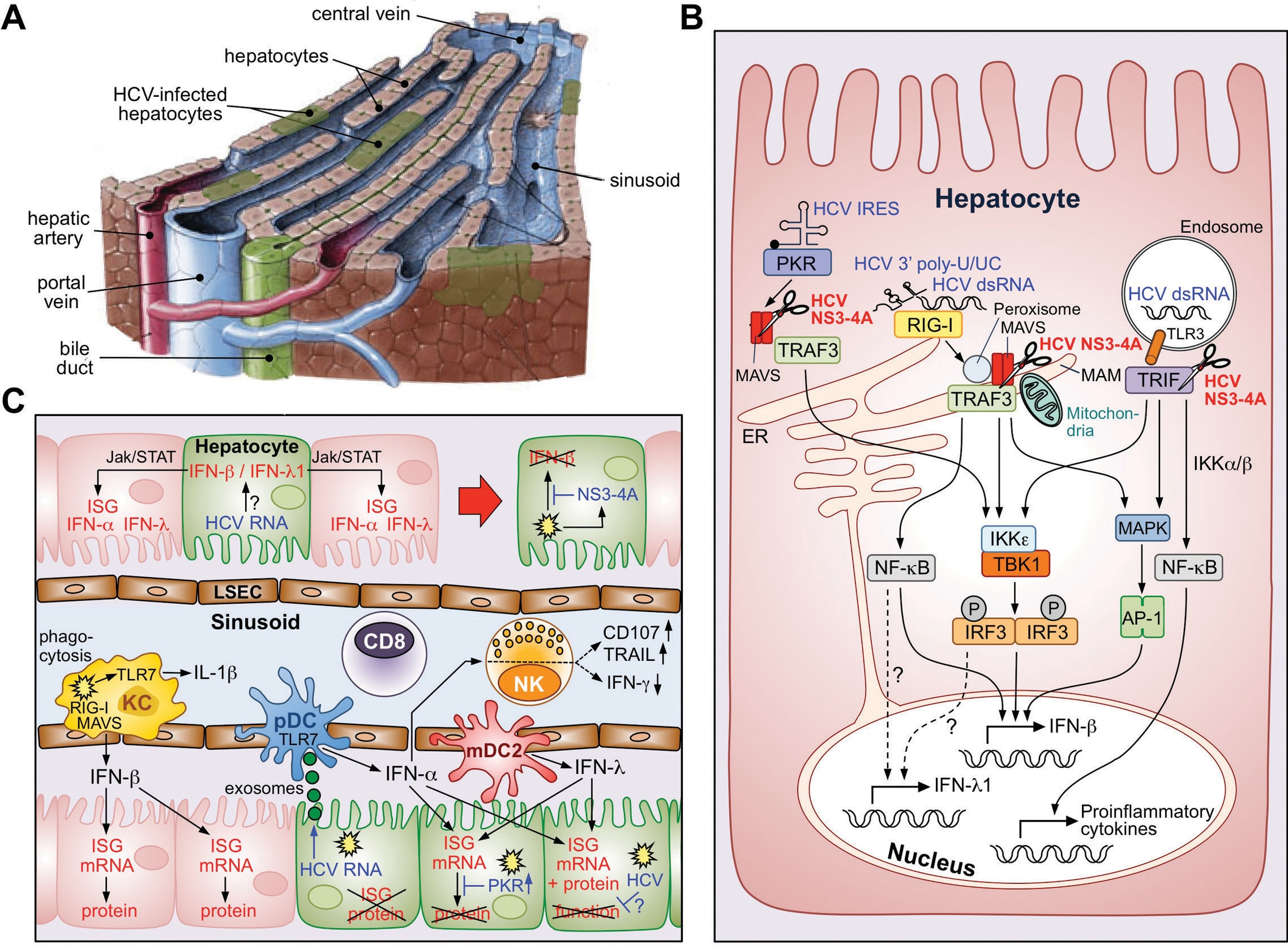
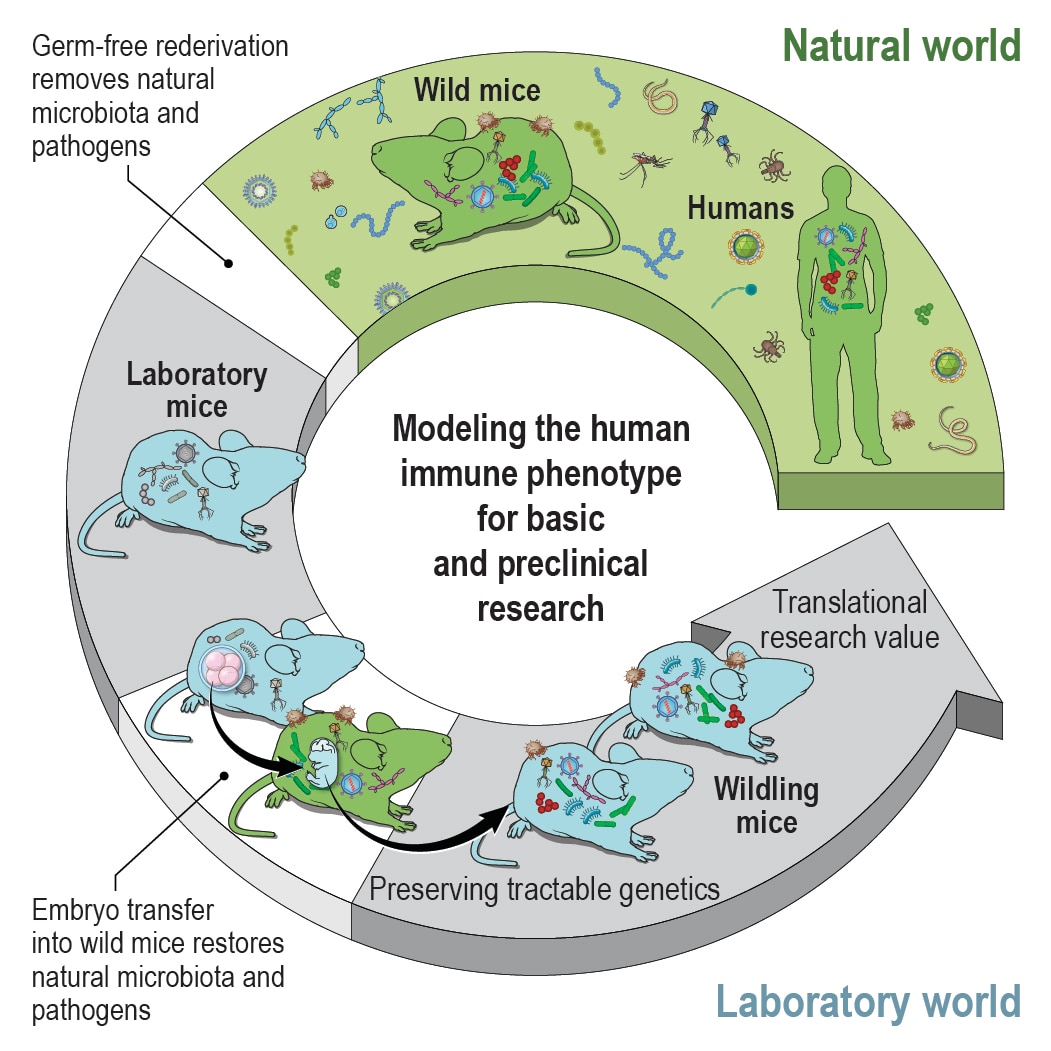 Due to its location and function the liver is constantly exposed to gut-derived microbial products. Bacterial translocation can cause life-threatening infections in end-stage cirrhosis. We previously reported innate-like lymphocytes (MAIT cells), which respond not only to inflammatory cytokines but also to metabolites from riboflavin-synthesizing gut bacteria, were depleted in advanced liver disease due to activation-induced apoptosis. Further, they exhibited an impaired response to stimulation with E. coli (Bolte et al., Gastroenterology 2017) that did not normalize after treatment and elimination of HCV, and thus may impact protection against bacterial infections.
Due to its location and function the liver is constantly exposed to gut-derived microbial products. Bacterial translocation can cause life-threatening infections in end-stage cirrhosis. We previously reported innate-like lymphocytes (MAIT cells), which respond not only to inflammatory cytokines but also to metabolites from riboflavin-synthesizing gut bacteria, were depleted in advanced liver disease due to activation-induced apoptosis. Further, they exhibited an impaired response to stimulation with E. coli (Bolte et al., Gastroenterology 2017) that did not normalize after treatment and elimination of HCV, and thus may impact protection against bacterial infections.
We currently take the following approaches:
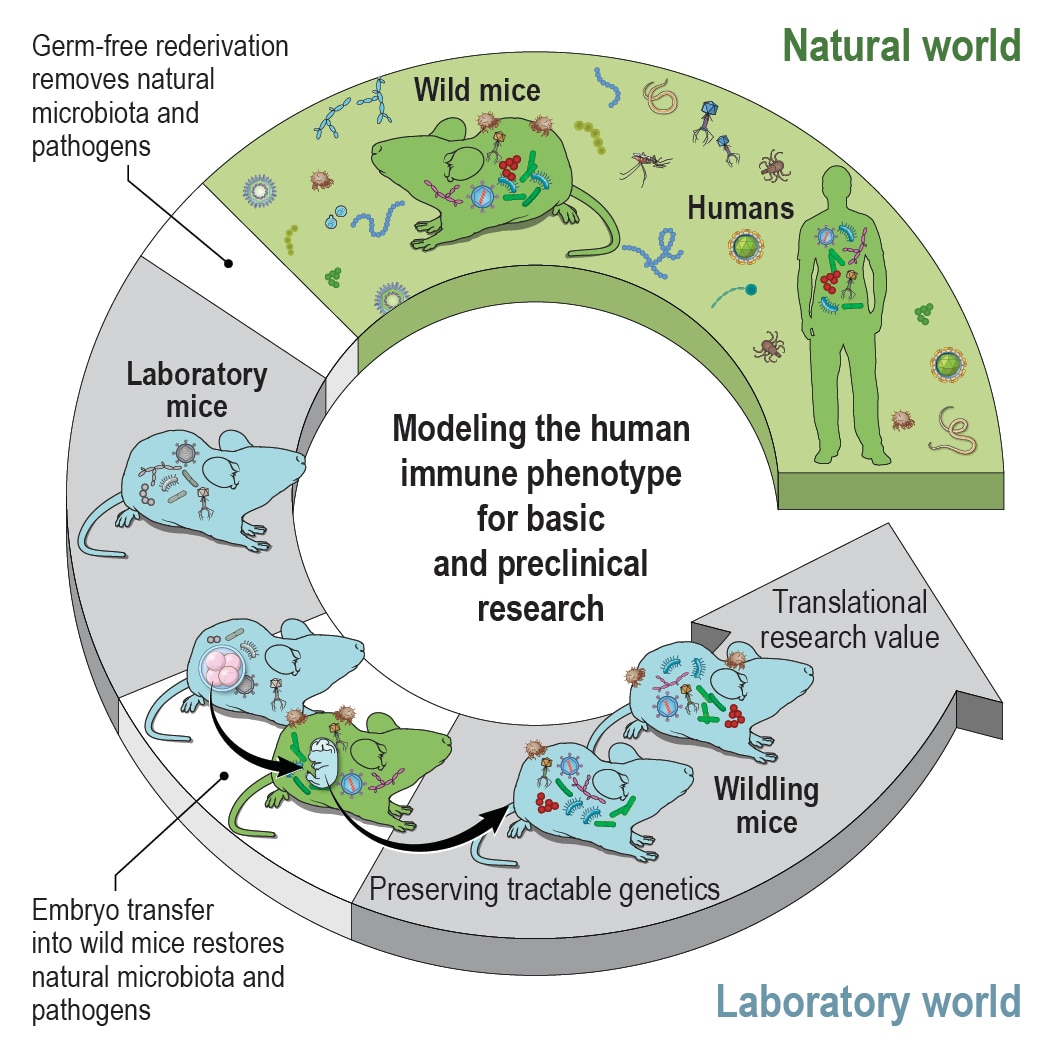
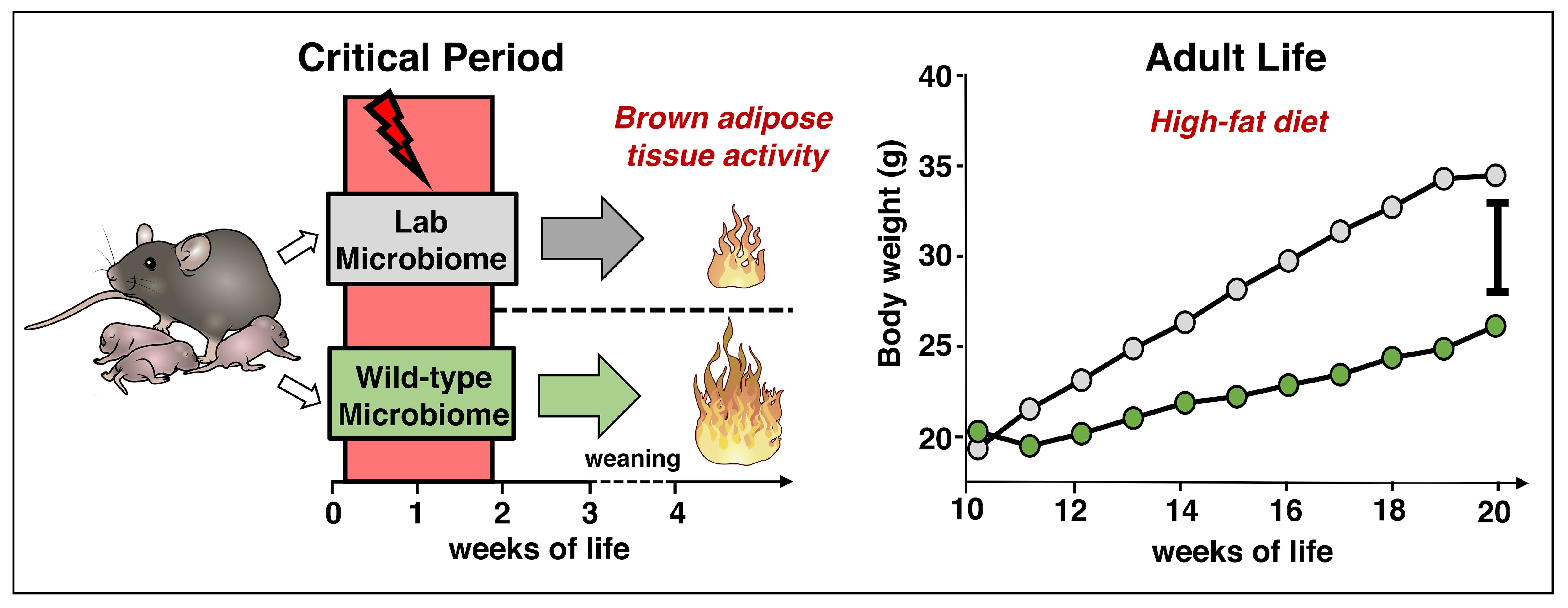
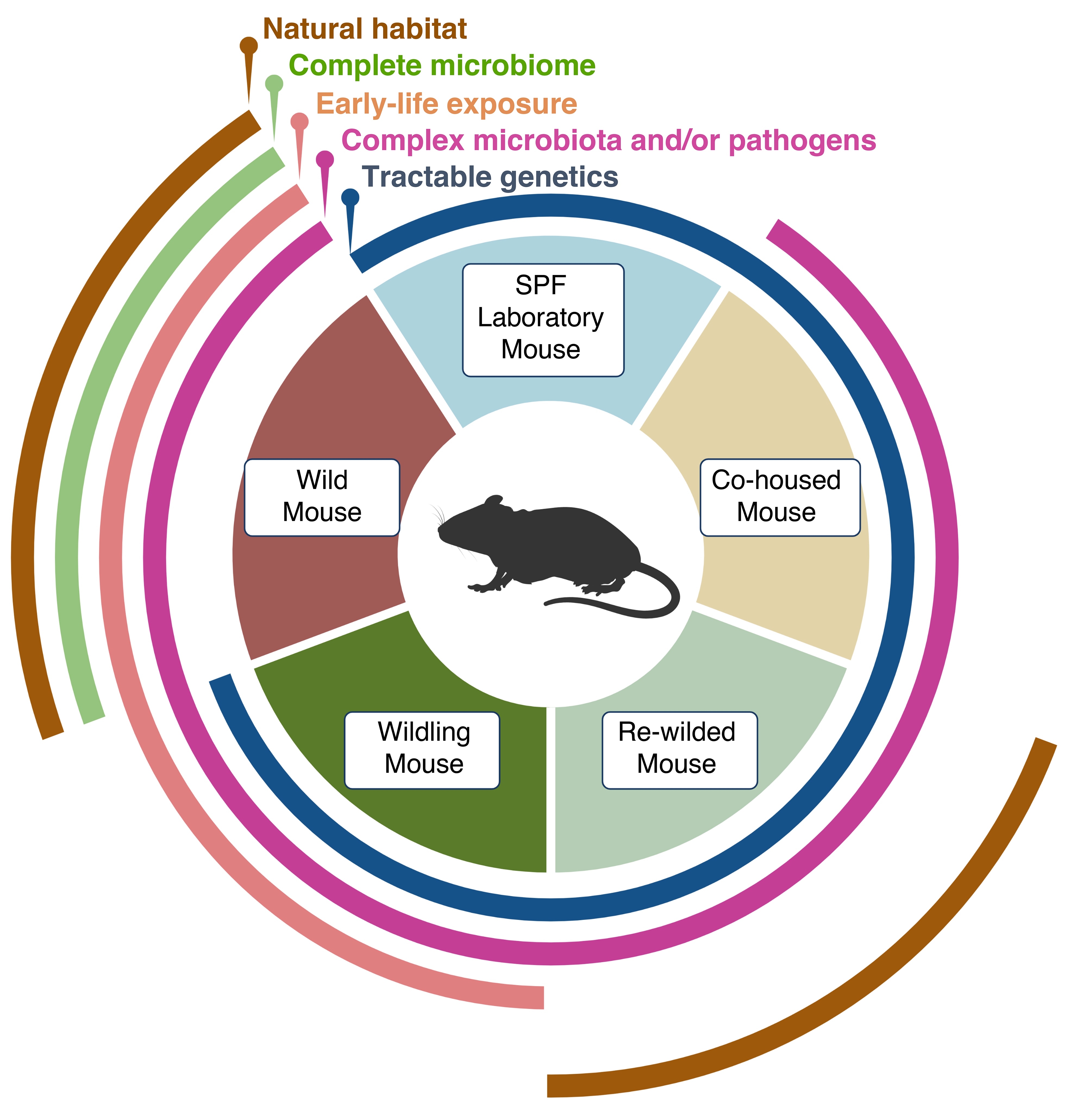
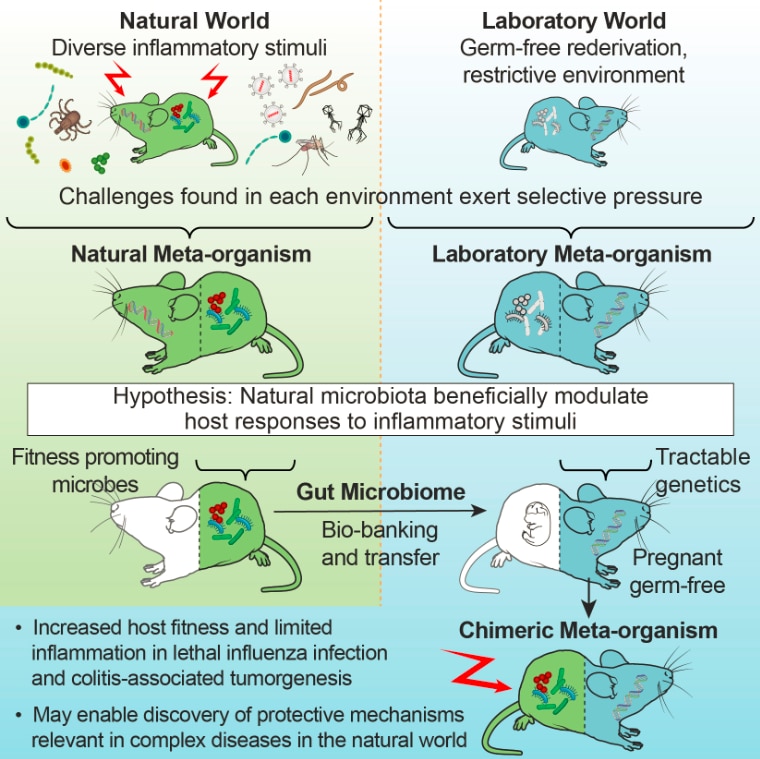
- To mechanistically evaluate the effects of the microbiome in inflammatory liver disease and metabolism we have established mouse models that combine the microbiome of wild mice with the tractable genetics of laboratory mice. This approach is based on the hypothesis that conventional laboratory mice lack microbiota/host interactions that are relevant in the natural world. These mice with natural microbiota had an immune profile that more closely resembled that of humans than that of conventional ‘clean’ laboratory mice. They predicted (as a preclinical model) human immune responses (in phase I studies) when conventional laboratory mice did not (Rosshart et al., Science 2019). We also demonstrated that early-life exposure to the wild mouse microbiome had metabolic effects through brown fat tissue activation inducing protection against diet induced obesity (Hild et al., Nature Metabolism 2021).
We are now using these mice to (i) discover disease-relevant mechanisms that are present in the natural world but absent in conventional lab mice, (ii) improve modelling of immune responses of humans in preclinical studies, and (iii) support drug development. - In a translational human immunology study, we are evaluating changes in microbiome and in inflammatory status during and after pregnancy. We characterize changes in the composition of the gut microbiome and metabolome in HBV-infected women and uninfected controls who are prospectively followed during and after pregnancy and to correlate these with innate and adaptive immune responses and hepatitis disease flares. The aim of this research is to understand the causative mechanisms, predictive markers and risk factors for hepatitis B disease flares during pregnancy.
Research in Plain Language
This research focuses on the body’s immune response to viruses that infect the liver and other organs. Using blood and liver biopsy samples from infected patients, we study the response of the infected cells themselves and the innate and adaptive immune cells that are called to the site of infection. We also analyze the mechanisms that viruses use as means to escape from immune responses, and the biological processes that result from chronic triggering of the immune response if the viruses persist. Finally, research on the gastrointestinal microbiome helps us better understand how immune responses and inflammation are regulated and may result in new approaches to regulate inflammation.
Research Images