About Our Research
The Liver Diseases Virology Section focuses on basic and translational research to understand the mechanisms of virus-related liver disease, to advance treatment and prevention of hepatitis B virus (HBV) and hepatitis C virus (HCV) infections, and to elucidate molecular pathways of viral hepatitis-associated hepatocellular carcinoma (HCC).
Research in Plain Language
Hepatitis B (HBV) and hepatitis C (HCV) infections affect more than 10 percent of the world population. They are the most common cause of liver disease including liver cancer—the fourth leading cause of cancer death. Current treatments for both viruses are effective but do not always work. While an effective vaccine for HBV is available, there is not one for HCV. In order to develop effective vaccines and better treatment approaches, we must understand how molecules, cells, and genes are altered over time in an infected person; how the virus interacts with the host; and how the immune system is affected by the virus. My laboratory focuses on these topics and pursue three approaches to study of HBV/HCV infections and the mechanisms that cause the disease.
Our first approach is to understand how antiviral treatments against HBV and HCV work and why they don’t work in some people. We explore the biological reasons that explain why some patients do not respond to treatment. Our second approach is to apply molecular, biochemical, and functional genomic tools to identify and characterize interactions between the virus, viral gene products, and the host. In the third approach, we generate new tools for developing and evaluating vaccines and antivirals by building and improving on currently available models.
Current Research
Hepatitis B and C viruses together infect more than 10% of the world population and are the most common cause of chronic liver disease and hepatocellular carcinoma, which is now the fourth leading cause of death from cancer in the world. Current therapies for HBV are less than optimal. Recent advances of direct-acting antivirals (DAAs) in HCV therapy have substantially improved the outlook for millions of HCV-infected people. The prospect for an HCV vaccine remains elusive. Given the global public health burden, it is crucial to improve on strategies to control the two infections including more affordable and effective therapies and preventive measures. To achieve this goal, we need to understand viral life cycle, viral mechanisms of productive and persistent infection, and virus-host interactions at the molecular, cellular and genomic levels. The thematic effort of my laboratory focuses on these topics.
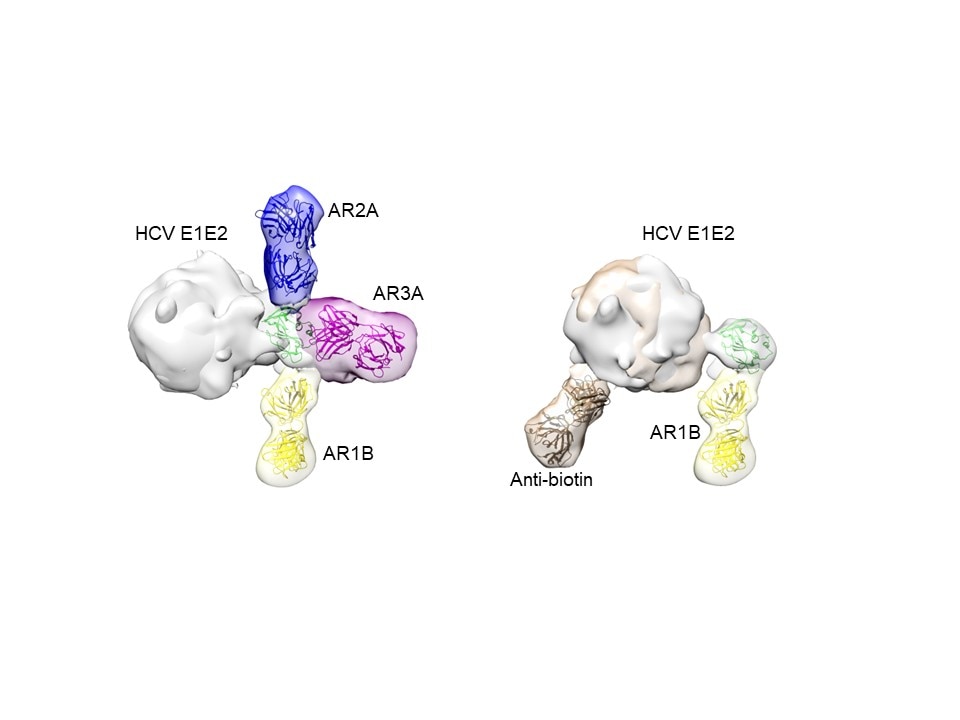
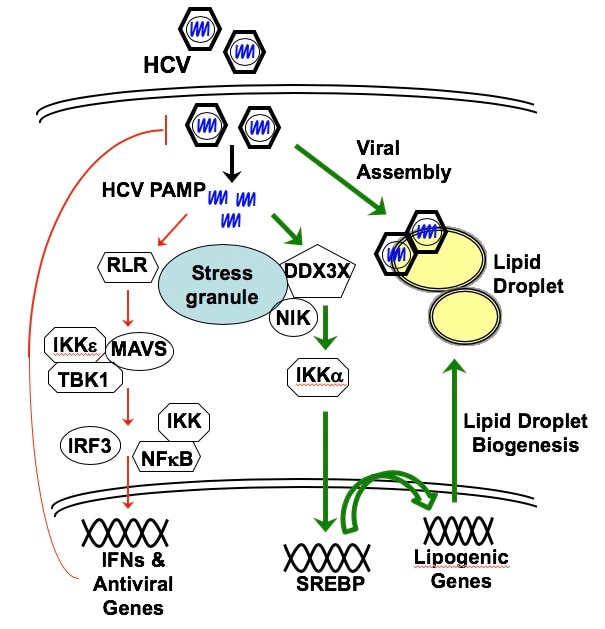
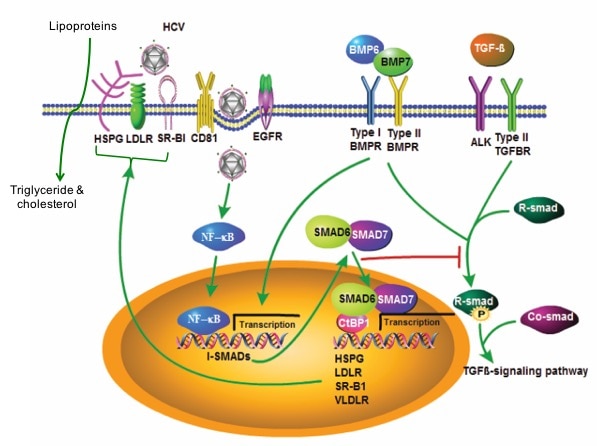
While many gaps in our knowledge of HCV still exist, great strides have been made in characterizing the virus and functions of viral genes as well as in unraveling the replication pathway and immunologic mechanisms of liver injury. HCV, like many other viruses, exploits host cellular machinery for productive infection. However, unlike other RNA viruses, HCV has a high propensity for persistent infection despite an active host immune response, which has been a major barrier for vaccine development. The virus commands unique mechanisms to counteract various host defenses. HCV gene products interact with many host factors and induce cellular alterations vital for viral replication, persistence and pathogenesis. The development of in vitro systems including infectious cell cultures affords the opportunity to fully characterize viral replication and virus-cell interactions as well as developing novel therapeutics. Animal models have also been developed to study HCV infection and pathogenesis in vivo.
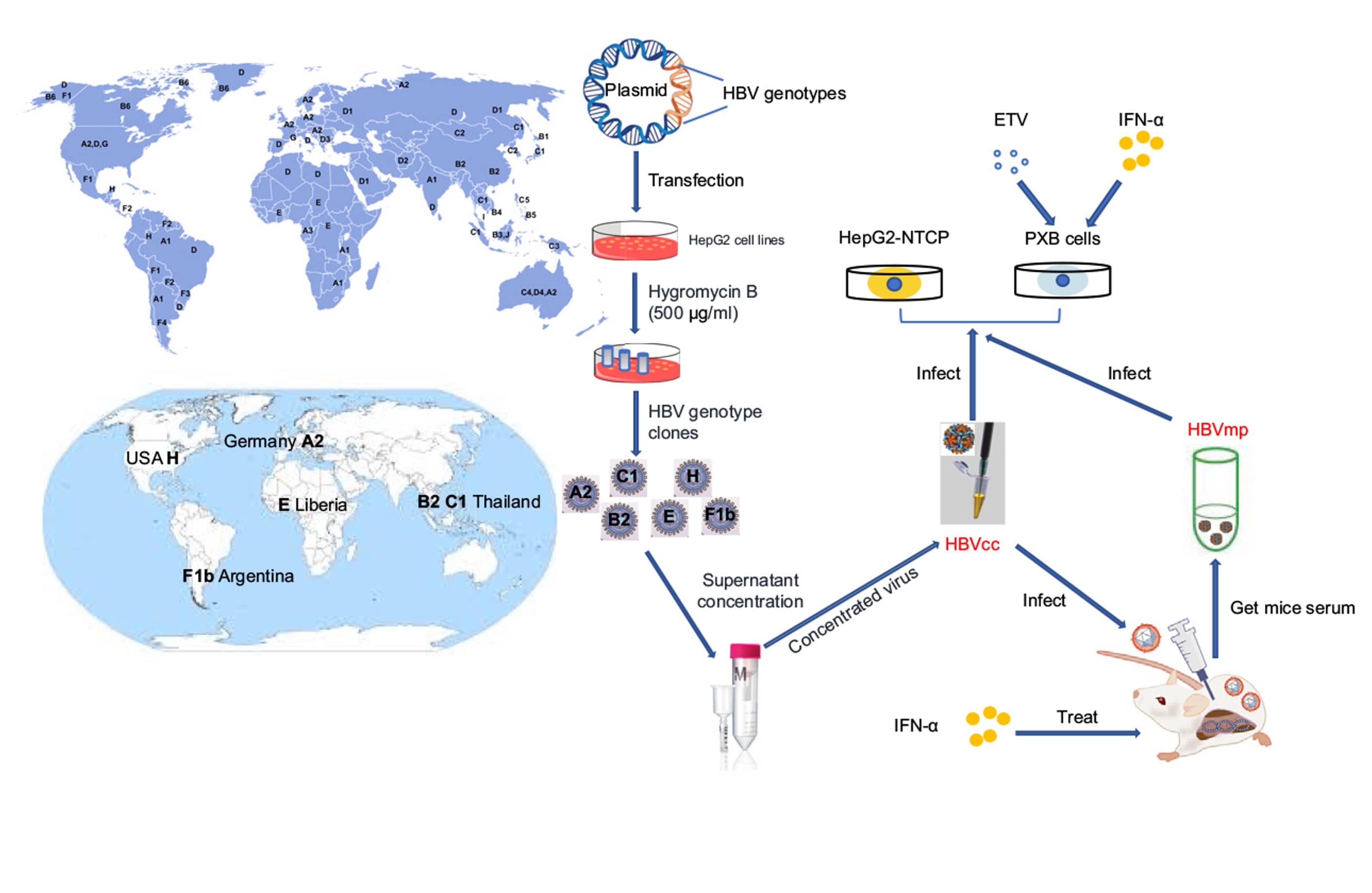
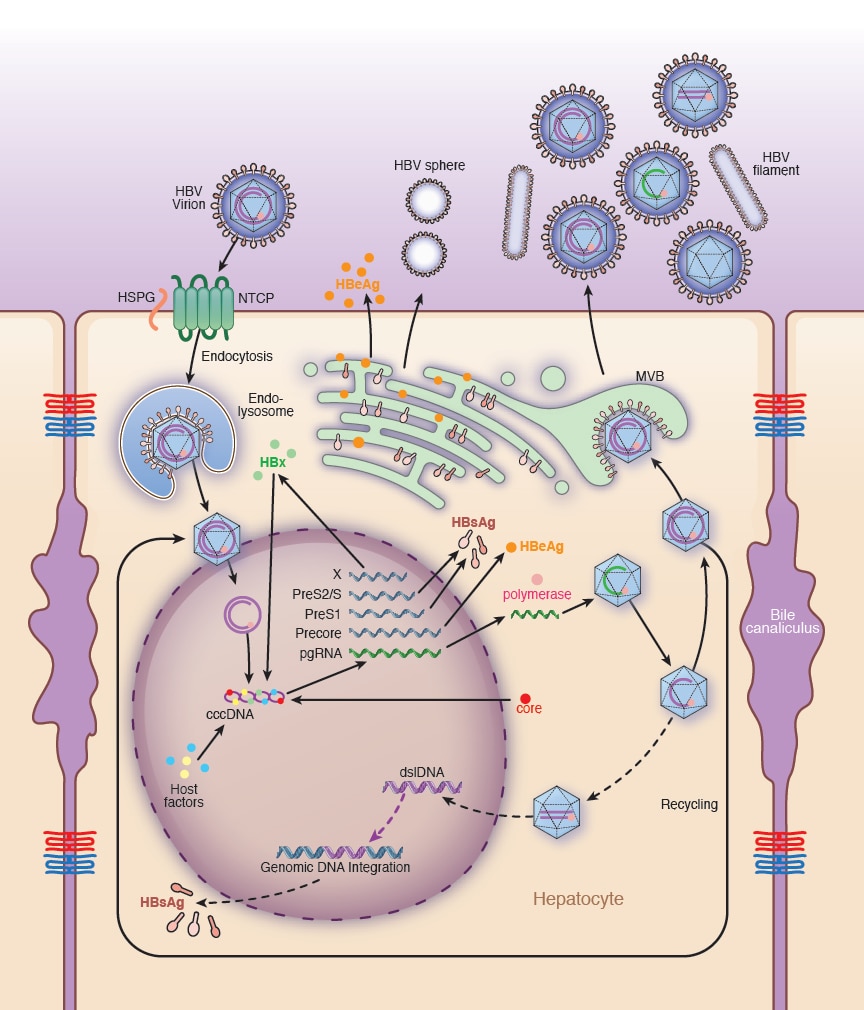
For HBV, while there are effective vaccines, therapies mostly suppress viral replication with little curative prospect. HBV replication and virus-host interactions have been studied extensively. The availability of HBV culture systems and small animal models has greatly advanced fundamental knowledge of the virus and the evaluation of vaccine and therapeutic candidates. The complexity of virus-host interactions and mechanism of persistence during HBV infection remains unclear.
Chronic infection with HBV or HCV is associated with development of hepatocellular carcinoma (HCC) that carries high morbidity and mortality and is difficult to diagnose and treat. Virus-associated HCC remains poorly understood and is an important unmet need in liver diseases.
Our three main approaches are
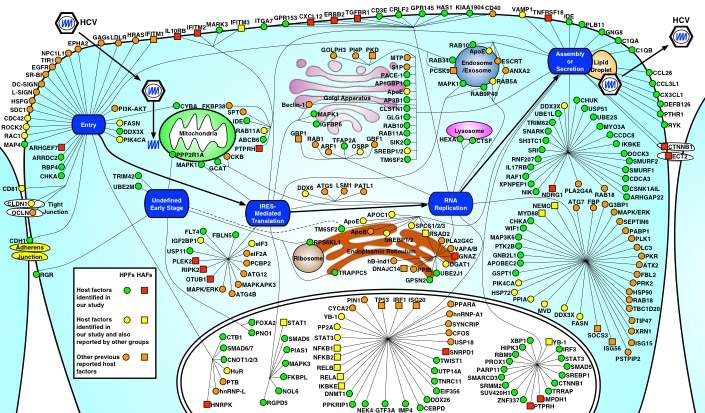
- To study virus-host interactions by applying genome-wide technologies and systems biology approaches.
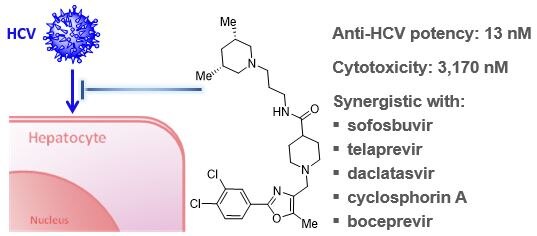
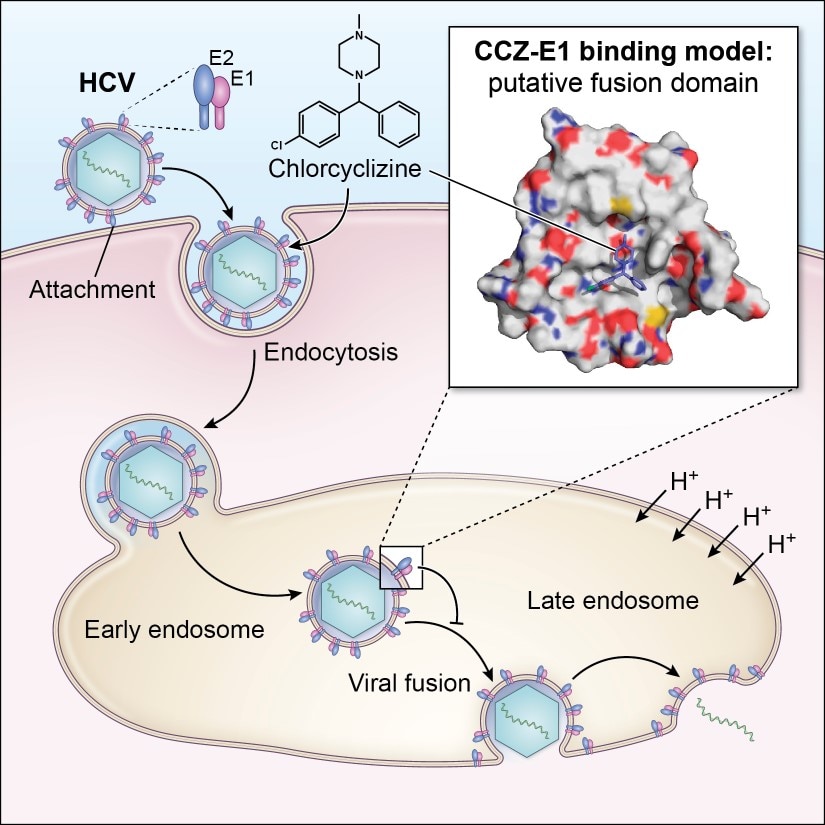
- To apply and develop technologies and model systems to discover novel small molecule probes of HBV and HCV life cycles and to explore them for potential therapeutic advances.
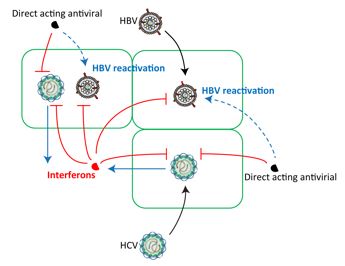
- To investigate the role of innate immunity and mechanisms of viral persistence in HBV and HCV pathogenesis and treatment response.
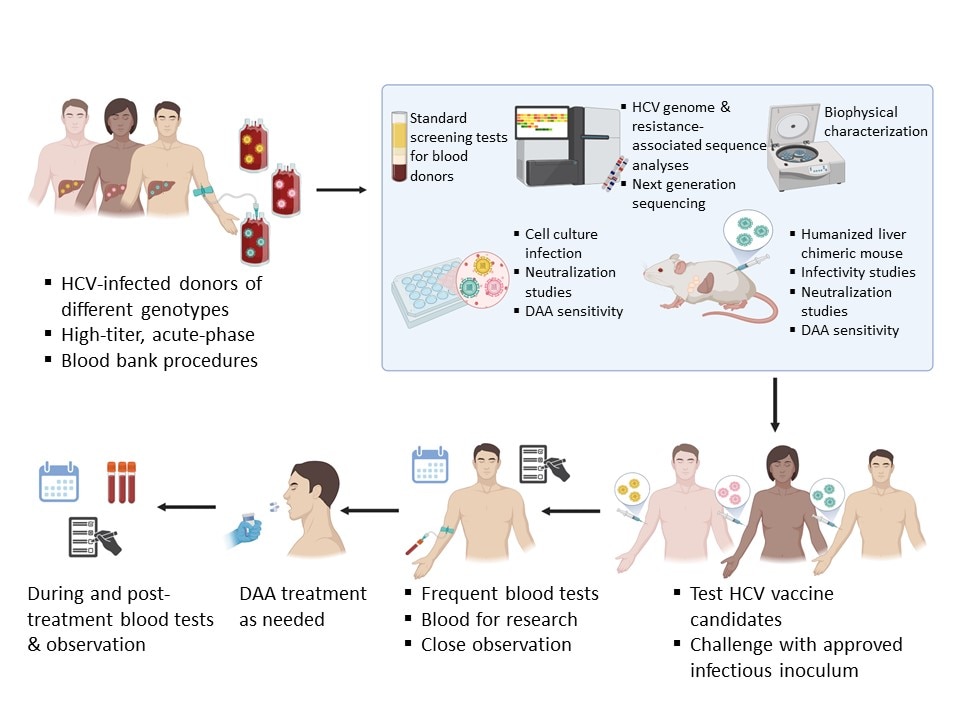
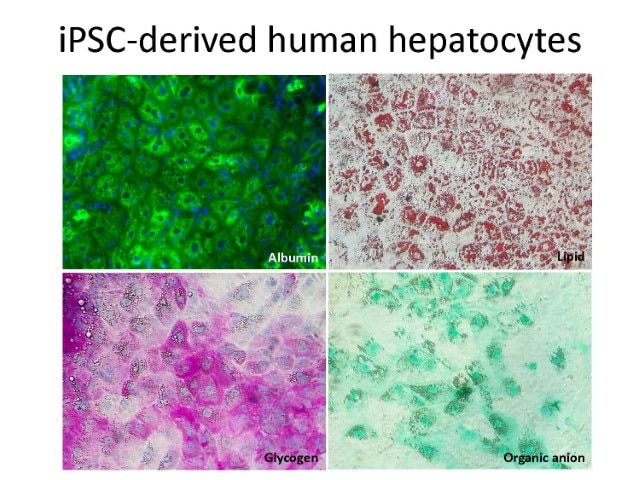
We successful adopted the technology of iPSC and hepatocyte differentiation as in vitro and in vivo models to study HBV and HCV infections. We demonstrated that these differentiated, hepatocyte-like cells (HLCs), despite an immature phenotype, are permissive to HCV infection in vitro and mount an interferon response. The HLCs could be engrafted in the liver parenchyma of immunodeficient mice. The HLCs can be maintained for an extended period of time in the liver of the chimeric mouse, where they undergo further maturation and proliferation. These chimeric mice were permissive to infection with HBV and HCV of different genotypes and support long-term infection.
We have also established an active research program on the increasingly important problem of hepatocellular carcinoma in the context of chronic viral hepatitis. We are developing various in vitro and in vivo models to identify and study potentially important procarcinogenic pathways that are triggered by HBV or HCV infection. By leveraging this knowledge, we hope to uncover novel strategies for preventive and therapeutic interventions of this deadly cancer.
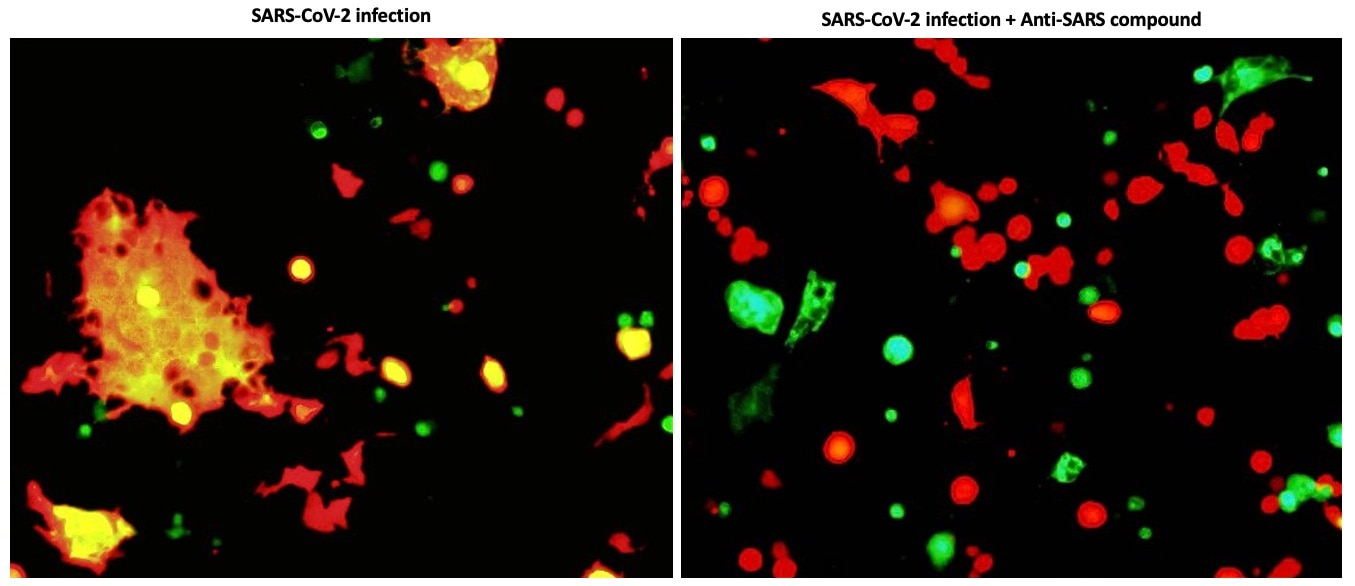
The recent coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has become a serious threat to global public health, underscoring the urgency to develop effective therapies. Therapeutics, and more specifically, direct-acting antiviral development, is still very much in its infancy. By leveraging our laboratory’s experience and expertise in antiviral development, we established a research program recently to identify and develop effective antivirals against the SARS-CoV-2, either by repurposing existing pharmaceuticals or developing new drugs. Many of the compounds we are developing may have broad-spectrum activities against many viruses including HCV and human coronaviruses.
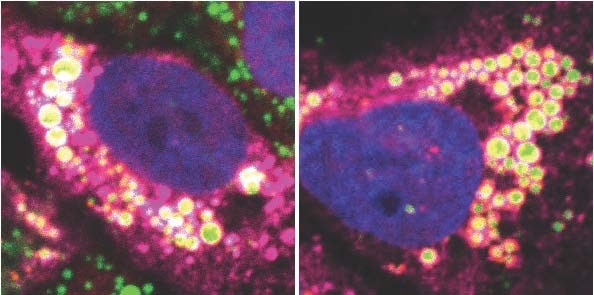
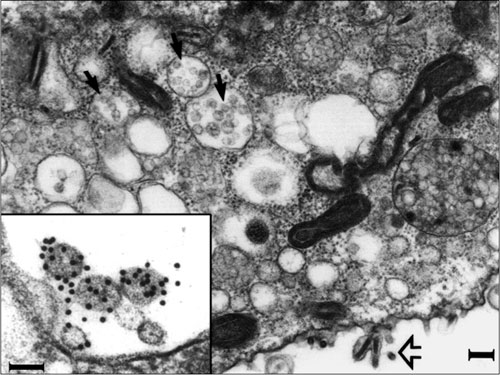
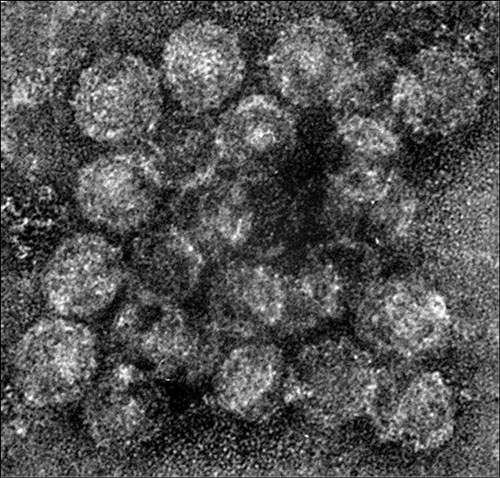
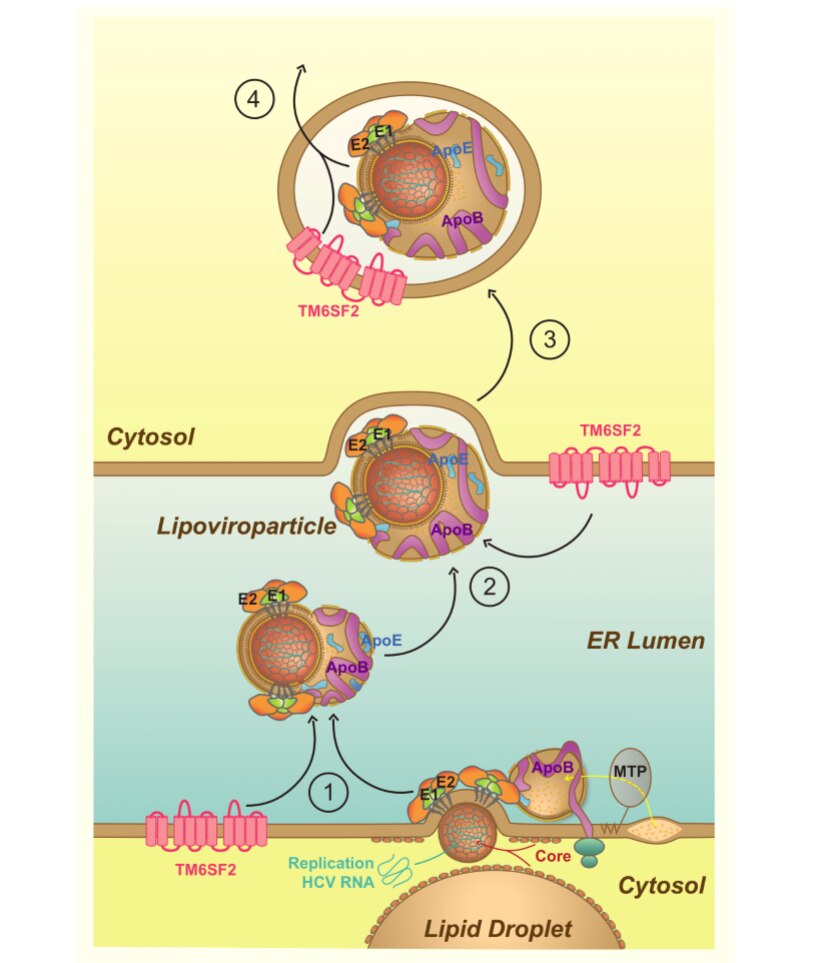
Research Images