About Our Research
Thyroid cancer is the most common endocrine malignancy. The incidence rate is rising faster than any other type of cancer, and it has a slowly increasing mortality rate. This phenomenon is likely due to the combination of two factors: (1) increase in imaging studies of the neck, leading to incidental discovery of thyroid nodules; (2) environmental factors, such as nuclear reactor accidents, and epidemy of obesity likely leading to a true increase in thyroid cancer incidence. It is important to differentiate an indolent disease course from a highly aggressive thyroid cancer to tailor therapeutic modalities towards avoiding over-treatment in low-risk patients and implementing effective targeted therapies for high-risk patients. Therefore, our scientific interest involves translational and clinical studies focused on both the optimization of thyroid nodules and thyroid cancer diagnosis, as well as appropriate risk stratification and therapy aimed at improving patients’ morbidity and mortality.
Current Research
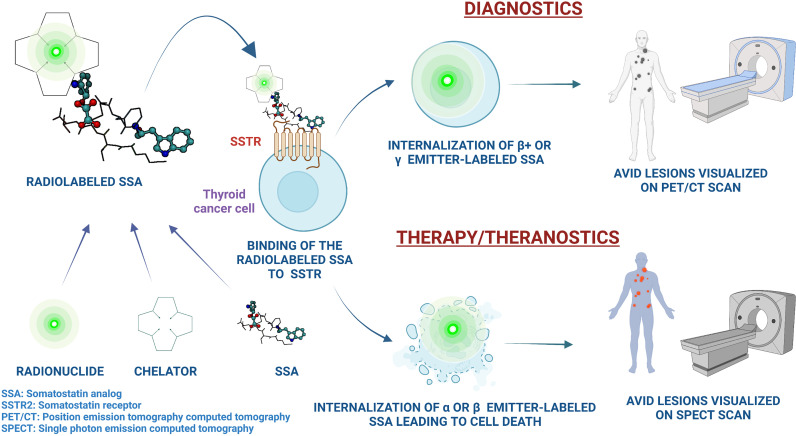
The mainstay of therapy for invasive, metastatic thyroid cancer consists of a total thyroidectomy followed by radioiodine therapy (RAI). We are focused on the optimization of RAI delivery to the target lesions to maximize its uptake and residence time in the tumor. Unfortunately, approximately 5-22% of patients become refractory to standard therapy with RAI. Moreover, certain subtypes of thyroid cancer are RAI-non-responsive, such as poorly differentiated, oncocytic (Hürthle cell) or C-cell-derived medullary thyroid cancer. Therefore, we are actively investigating somatostatin receptors and other molecular targets to test the role of new radiolabeled analogs in the imaging and treatment of metastatic radioactive iodine non-avid thyroid cancer and medullary thyroid cancer.
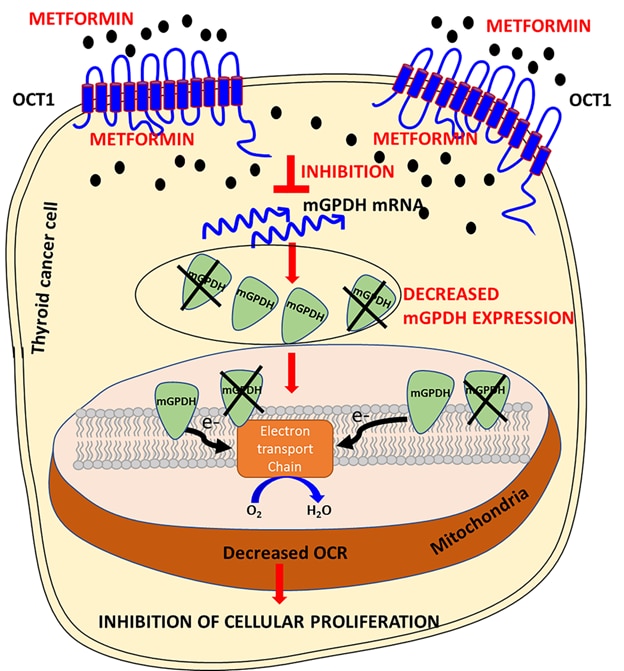
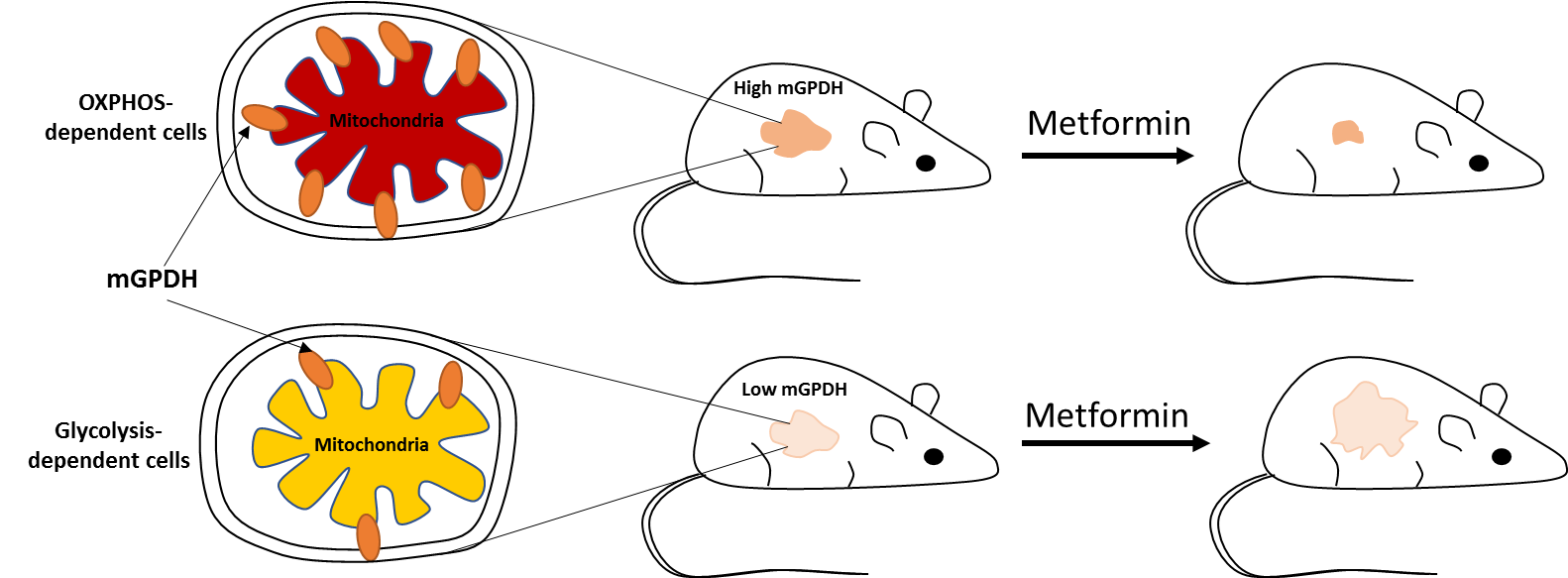
We also documented that treatment with an anti-diabetic drug metformin is associated with smaller tumor size and higher remission rate in diabetic patients with thyroid cancer. We described the in vitro mechanisms of metformin action in differentiated and medullary thyroid cancer and documented the role of the microenvironment in the modulation of the sensitivity to metformin. We have shown that one of the mechanisms of metformin action is inhibition of oxidative phosphorylation, leading to a change in the metabolic profile of cancer cells and in energetic stress. We found that mitochondrial glycerophosphate dehydrogenase (mGPDH) is one of the metformin targets in thyroid cancer. We are investigating a molecular and metabolic signature of thyroid cancer, which could be targeted by metformin and other agents affecting cancer metabolism.
Applying our Research
The knowledge gained on the cross-talk between the cancer signaling pathways and cell metabolism from our preclinical models, as well as identification of new molecular targets for imaging and treatment, will form a basis for targeted therapies and clinical trials in the most challenging group of thyroid cancer patients – with RAI non-avid disease and metastatic medullary thyroid cancer.
Need for Further Study
The ultimate goal of our group is to translate our preclinical data from the bench to the bedside.
Research Images